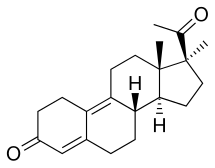
Demegestone
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Lutionex |
| Other names | Dimegestone; R-2453; RU-2453; 17α-Methyl-δ9-19-norprogesterone; 17α-Methyl-19-norpregna-4,9-diene-3,20-dione |
| Routes of administration | By mouth[1] |
| Drug class | Progestogen; Progestin |
| ATC code | |
| Pharmacokinetic data | |
| Bioavailability | Good[2] |
| Metabolism | Hydroxylation, others[2] |
| Metabolites | • 21-Hydroxydemegestone[2] • Others[2] |
| Excretion | Urine[2] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.030.278 |
| Chemical and physical data | |
| Formula | C21H28O2 |
| Molar mass | 312.453 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Demegestone, sold under the brand name Lutionex, is a progestin medication which was previously used to treat luteal insufficiency but is now no longer marketed.[3][4][5][6][7] It is taken by mouth.[2][1]
Demegestone is a progestin, or a synthetic progestogen, and hence is an agonist of the progesterone receptor, the biological target of progestogens like progesterone.[6][2][8] It has no androgenic activity.[2]
Demegestone was first described in 1966 and was introduced for medical use in France in 1974.[3][4] It has only been marketed in France, and has since been discontinued in this country.[5][4]
Medical uses
[edit]Demegestone has been used to treat luteal insufficiency.[7] It has also been studied in combination with estrogens, such as moxestrol, as an oral contraceptive and treatment for infertility.[1][9][10]
Side effects
[edit]Pharmacology
[edit]Pharmacodynamics
[edit]Demegestone is a progestogen, and hence is an agonist of the progesterone receptor (PR).[6][8][2] It is a highly potent progestogen, showing 50 times the potency of progesterone in the Clauberg test.[2] The ovulation-inbhiting dosage of demegestone is 2.5 mg/day, while the endometrial transformation dosage is 100 mg per cycle.[11] The medication is devoid of androgenic activity,[2] and instead has some antiandrogenic activity.[12] Demegestone has low affinity for the glucocorticoid receptor.[13] In a particular bioassay, both demegestone and progesterone showed antiglucocorticoid rather than glucocorticoid activity.[14] The major metabolite of demegestone, a 21-hydroxylated metabolite, is a moderately potent progestogen (4 times the potency of progesterone) and a weak mineralocorticoid (2% of the potency of deoxycorticosterone).[2]
| Compound | PR | AR | ER | GR | MR | SHBG | CBG | |
|---|---|---|---|---|---|---|---|---|
| Demegestone | 230 | 1 | 0 | 5 | 1–2 | ? | ? | |
| Notes: Values are percentages (%). Reference ligands (100%) were progesterone for the PR, testosterone for the AR, E2 for the ER, DEXA for the GR, aldosterone for the MR, DHT for SHBG, and cortisol for CBG. Sources: [13][15][16][17] | ||||||||
Pharmacokinetics
[edit]Demegestone has good bioavailability.[2] The initial volume of distribution of demegestone is 31 L.[2] Demegestone is metabolized by hydroxylation at the C21, C1, C2, and C11 positions, which is eventually followed by A-ring aromatization after 1,2-dehydration.[2] The major metabolite of demegestone is a 21-hydroxy derivative.[2] The metabolic clearance rate of demegestone is 20 L/h.[2] Its biological half-lives are 2.39 and 0.24 hours with intravenous injection.[2] Demegestone and/or its metabolites are excreted, at least in part, in urine.[2]
Chemistry
[edit]Demegestone, also known as 17α-methyl-δ9-19-norprogesterone or as 17α-methyl-19-norpregna-4,9-diene-3,20-dione, is a synthetic norpregnane steroid and a derivative of progesterone.[3][4][6] It is specifically a combined derivative of 17α-methylprogesterone and 19-norprogesterone, or of 17α-methyl-19-norprogesterone.[3][4][6] Related derivatives of 17α-methyl-19-norprogesterone include promegestone and trimegestone.[3][6]
History
[edit]Demegestone was first described in the literature in 1964 and was introduced for medical use in 1974 in France.[3][4] It was developed by Roussel Uclaf.[4]
Society and culture
[edit]Generic names
[edit]Demegestone is the generic name of the drug and its INN.[3] It is also known by its developmental code name R-2453 or RU-2453.[3]
Brand names
[edit]Demegestone was marketed under the brand name Lutionex.[3][4]
Availability
[edit]Demegestone is no longer marketed and hence is no longer available in any country.[5] It was previously available in France.[5][4]
References
[edit]- ^ a b c Iizuka R, Hayashi M, Kamouchi Y, Yamanaka K (1971). "Evaluation of a low-dose progestagen as a contraceptive". Nihon Funin Gakkai Zasshi. 16 (1): 68–82. PMID 12158578.
- ^ a b c d e f g h i j k l m n o p q r s Raynaud JP, Cousty C, Salmon J (1974). "121. Metabolic studies of R2453, a highly potent progestin". Journal of Steroid Biochemistry. 5 (4): 324. doi:10.1016/0022-4731(74)90266-0. ISSN 0022-4731.
- ^ a b c d e f g h i Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 356–. ISBN 978-1-4757-2085-3.
- ^ a b c d e f g h i William Andrew Publishing (22 October 2013). Pharmaceutical Manufacturing Encyclopedia (3rd ed.). Elsevier. pp. 1215–. ISBN 978-0-8155-1856-3.
- ^ a b c d "Demegestone". Micromedex.: [permanent dead link]
- ^ a b c d e f Kuhl H (August 2005). "Pharmacology of estrogens and progestogens: influence of different routes of administration". Climacteric. 8 (Suppl 1): 3–63. doi:10.1080/13697130500148875. PMID 16112947. S2CID 24616324.
- ^ a b Pugeat M, Lejeune H, Dechaud H, Brébant C, Mallein R, Tourniaire J (1988). "[Luteal insufficiency and elevation of sex-binding proteins by demegestone]". Revue Française de Gynécologie et d'Obstétrique (in French). 83 (7–9): 495–498. PMID 3194612.
- ^ a b Lee DL, Kollman PA, Marsh FJ, Wolff ME (September 1977). "Quantitative relationships between steroid structure and binding to putative progesterone receptors". Journal of Medicinal Chemistry. 20 (9): 1139–1146. doi:10.1021/jm00219a006. PMID 926114.
- ^ Hamada H, Nagao H, Toyoda H, Hayashi H, Akihiro L, Kotaki S (1970). "[Clinical observation on oral contraceptive effect by R-2453 (Abstracts of Papers Presented at Showa 44 in the field of gynecology])". Japanese Journal of Obstetrics and Gynecology-Acta Obstetrica et Gynaecologica Japonica. 22 (7): 753.
- ^ Levrier M (January 1979). "Treatment of Ovarian Sterility with Combined Moxestrol-Demegestone Preparation". Journal de Gynécologie Obstétrique et Biologie de la Reproduction. 8 (1). Paris, France: Masson Editeur: 89.
- ^ Rabe T, Goeckenjan M, Ahrendt HJ, Crosignani PG, Dinger JC, Mueck AO, et al. (October 2011). "Oral Contraceptive Pills: Combinations, Dosages and the Rationale behind 50 Years or Oral Hormonal Contraceptive Development" (PDF). Journal für Reproduktionsmedizinund Endokrinologie. 8 (1): 58–129.
- ^ Raynaud JP, Ojasoo T, Labrie F (1981). "Steroid hormones—agonists and antagonists". Mechanisms of Steroid Action. Macmillan Education UK. pp. 145–158. doi:10.1007/978-1-349-81345-2_11. ISBN 978-1-349-81347-6.
- ^ a b Delettré J, Mornon JP, Lepicard G, Ojasoo T, Raynaud JP (January 1980). "Steroid flexibility and receptor specificity". Journal of Steroid Biochemistry. 13 (1): 45–59. doi:10.1016/0022-4731(80)90112-0. PMID 7382482.
- ^ Dausse JP, Duval D, Meyer P, Gaignault JC, Marchandeau C, Raynaud JP (September 1977). "The relationship between glucocorticoid structure and effects upon thymocytes". Molecular Pharmacology. 13 (5): 948–955. PMID 895725.
- ^ Raynaud JP, Bouton MM, Moguilewsky M, Ojasoo T, Philibert D, Beck G, et al. (January 1980). "Steroid hormone receptors and pharmacology". Journal of Steroid Biochemistry. 12: 143–157. doi:10.1016/0022-4731(80)90264-2. PMID 7421203.
- ^ Ojasoo T, Raynaud JP, Doé JC (January 1994). "Affiliations among steroid receptors as revealed by multivariate analysis of steroid binding data". The Journal of Steroid Biochemistry and Molecular Biology. 48 (1): 31–46. doi:10.1016/0960-0760(94)90248-8. PMID 8136304. S2CID 21336380.
- ^ Ojasoo T, Raynaud JP (November 1978). "Unique steroid congeners for receptor studies". Cancer Research. 38 (11 Pt 2): 4186–4198. PMID 359134.
