User:Abhinav2009/Phosphofructokinase 1
| 6-phosphofructokinase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 | |||||||||
| Identifiers | |||||||||
| EC no. | 2.7.1.11 | ||||||||
| CAS no. | 9001-80-3 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
| Phosphofructokinase | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||||
| Symbol | PFK | ||||||||||
| Pfam | PF00365 | ||||||||||
| Pfam clan | CL0240 | ||||||||||
| InterPro | IPR000023 | ||||||||||
| PROSITE | PDOC00336 | ||||||||||
| SCOP2 | 5pfk / SCOPe / SUPFAM | ||||||||||
| |||||||||||
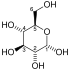
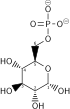
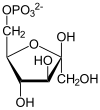
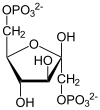
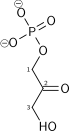
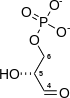
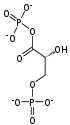
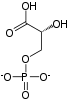
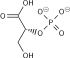
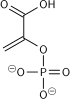
Phosphofructokinase-1 (PFK-1) is one of the most important regulatory enzymes (EC 2.7.1.11) of glycolysis. It is an allosteric enzyme made of 4 subunits and controlled by many activators and inhibitors. PFK-1 catalyzes the important "committed" step of glycolysis, the conversion of fructose 6-phosphate and ATP to fructose 1,6-bisphosphate and ADP.
| β-D-fructose 6-phosphate | {{{forward_enzyme}}} | β-D-fructose 1,6-bisphosphate | |
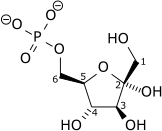
|
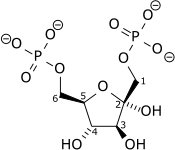
| ||
| {{{minor_forward_substrate(s)}}} | {{{minor_forward_product(s)}}} | ||
| [[image:Biochem_reaction_arrow_{{{reaction_direction_(forward/reversible/reverse)}}}_NNYY_horiz_med.svg|75px]] | |||
| Pi | H2O | ||
| Fructose bisphosphatase | |||
Structure
[edit]Mammalian PFK1 is a 340kd[1] tetramer composed of three types of subunit: muscle (M), liver (L), and platelet (P). The composition of the PFK1 tetramer differs according to the tissue type it is present in. For example, mature muscle expresses only the M isozyme, therefore, the muscle PFK1 is composed solely of homotetramers of M4. The liver and kidneys express predominantly the L isoform. Erythrocytes express both M and L subunits which randomly tetramerize to form M4, L4 and the three hybrid forms of the enzyme (ML3, M2L2, M3L). As a result, the kinetic and regulatory properties of the various isoenzymes pools are dependent on subunit composition. Tissue-specific changes in PFK activity and isoenzymic content contribute significantly to the diversities of glycolytic and gluconeogenic rates which have been observed for different tissues.[2]
PFK1 is an allosteric enzyme and has a structure similar to that of hemoglobin insofar as it is a dimer of a dimer.[3] One half of each dimer contains the ATP binding site whereas the other half the substrate (fructose-6-phosphate or (F6P)) binding site as well as a separate allosteric binding site.[4]
Mechanism
[edit]PFK1 is an allosteric enzyme whose activity can be described using the symmetry model of allosterism[5] whereby there is a concerted transition from an enzymatically inactive T-state to the active R-state. F6P binds with a high affinity to the R state but not the T state enzyme. For every molecule of F6P that binds to PFK1, the enzyme progressively shifts from T state to the R state. Thus a graph plotting PFK1 activity against increasing F6P concentrations would adopt the sigmoidal curve shape traditionally associated with allosteric enzymes.
PFK1 belongs to the family of phosphotransferases and it catalyzes the transfer of γ-phosphate from ATP to fructose-6-phosphate. The PFK1 active site comprises both the ATP-Mg2+ and the F6P binding sites. Some proposed residues involved with substrate binding in E. coli FPK1 include Asp127 and Arg171.[6] In B. stearothermophilus PFK1, the positively charged side chain of Arg162 residue forms a hydrogen-bonded salt bridge with the negatively charged phosphate group of F6P, an interaction which stabilizes the R state relative to the T state and is partly responsible for the homotropic effect of F6P binding. In the T state, enzyme conformation shifts slightly such that the space previously taken up by the Arg162 is replaced with Glu161. This swap in positions between adjacent amino acid residues inhibits the ability of F6P to bind the enzyme.
Allosteric activators such as AMP and ADP bind to the allosteric site as to facilitate the formation of the R state by inducing structural changes in the enzyme. Similarly, inhibitors such as ATP and PEP bind to the same allosteric site and facilitate the formation of the T state, thereby inhibiting enzyme activity.
Regulation
[edit]PFK1 is the most important control site in the mammalian glycolytic pathway. This step is subject to extensive regulation since it is not only highly endergonic under physiological conditions, but also because it is a committed step - the first irreversible reaction unique to the glycolytic pathway. This leads to a precise control of glucose and the other monosaccharides galactose and fructose going down the glycolytic pathway. Before this enzyme's reaction, glucose-6-phosphate can potentially travel down the pentose phosphate pathway, or be converted to glucose-1-phosphate for glycogenesis.
PFK1 is allosterically inhibited by high levels of ATP but AMP reverses the inhibitory action of ATP. Therefore, the activity of the enzyme increases when the cellular ATP/AMP ratio is lowered. Glycolysis is thus stimulated when energy charge falls. PFK1 has two sites with different affinities for ATP which is both a substrate and an inhibitor.[1]
PFK1 is also inhibited by low pH levels which augment the inhibitory effect of ATP. The pH falls when muscle is functioning anaerobically and producing excessive quantities of lactic acid. This inhibitory effect serves to protect the muscle from damage that would result from the accumulation of too much acid.[1]
Finally, PFK1 is allosterically inhibited by both PEP and citrate. Phosphoenolpyruvic acid is a product further downstream the glycolytic pathway and citrate is an early intermediate in the citric acid cycle. Citrate buildup is a sign of the citric acid cycle reaching saturation and thus glycolysis slows down as there is no longer any need to commit more glucose to degradation.
PFK1 is allosterically activated by a high concentration of AMP, but the most potent activator is fructose 2,6-bisphosphate, which is also produced from fructose-6-phosphate by PFK2. Hence, an abundance of F6P results in a higher concentration of fructose 2,6-bisphosphate (F-2,6-BP). The binding of F-2,6-BP increases the affinity of PFK1 for F6P and diminishes the inhibitory effect of ATP. This is an example of feedforward stimulation as glycolysis is accelerated when glucose is abundant.[1]
PFK is inhibited by glucagon through repression of synthesis. Glucagon activates protein kinase A which, in turn, shuts off the kinase activity of PFK2. This reverses any synthesis of F-2,6-BP from F6P and thus inhibits PFK1 activity.
The precise regulation of PFK1 prevents glycolysis and gluconeogenesis from occurring simultaneously. However, there is substrate cycling between F6P and F-1,6-BP. Fructose-1,6-bisphosphatase (FBPase) catalyzes the hydrolysis of F-1,6-BP back to F6P, the reverse reaction catalyzed by PFK1. There is a small amount of FBPase activity during glycolysis and some PFK1 activity during gluconeogenesis. This cycle allows for the amplification of metabolic signals as well as the generation of heat by ATP hydrolysis.
Genes
[edit]There are three phosphofructokinase genes in humans:
Disease
[edit]A genetic mutation in the PFKM gene results in Tarui's disease, a glycogen storage disease where the ability of certain cell types to utilize carbohydrates as a source of energy is impaired.[7]
See also
[edit]- PFK2 (converts fructose 6-phosphate to fructose 2,6-bisphosphate through on site, or the opposite, on another site)
- PFP (reversibly interconverts fructose 6-phosphate and fructose 1,6-bisphosphate using inorganic pyrophosphate rather than ATP)
- fructose bisphosphatase (hydrolyses fructose 1,6-bisphosphate to fructose 6-phosphate)
References
[edit]- ^ a b c d Stryer, Lubert; Berg, Jeremy Mark; Tymoczko, John L. (2007). Biochemistry (Sixth ed.). San Francisco: W.H. Freeman. ISBN 978-0-7167-8724-2.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ Dunaway GA, Kasten TP, Sebo T, Trapp R (1988). "Analysis of the phosphofructokinase subunits and isoenzymes in human tissues". Biochem. J. 251 (3): 677–83. doi:10.1042/bj2510677. PMC 1149058. PMID 2970843.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ PDB: 4pfk; Evans PR, Farrants GW, Hudson PJ (1981). "Phosphofructokinase: structure and control". Philos. Trans. R. Soc. Lond., B, Biol. Sci. 293 (1063): 53–62. doi:10.1098/rstb.1981.0059. PMID 6115424.
{{cite journal}}: Unknown parameter|lay-source=ignored (help); Unknown parameter|lay-url=ignored (help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Shirakihara Y, Evans PR (1988). "Crystal structure of the complex of phosphofructokinase from Escherichia coli with its reaction products". J. Mol. Biol. 204 (4): 973–94. doi:10.1016/0022-2836(88)90056-3. PMID 2975709.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Peskov K, Goryanin I, Demin O (2008). "Kinetic model of phosphofructokinase-1 from Escherichia coli". J Bioinform Comput Biol. 6 (4): 843–67. doi:10.1142/S0219720008003643. PMID 18763746.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Hellinga HW, Evans PR (1987). "Mutations in the active site of Escherichia coli phosphofructokinase". Nature. 327 (6121): 437–9. doi:10.1038/327437a0. PMID 2953977.
- ^ Nakajima H, Raben N, Hamaguchi T, Yamasaki T (2002). "Phosphofructokinase deficiency; past, present and future". Curr. Mol. Med. 2 (2): 197–212. doi:10.2174/1566524024605734. PMID 11949936.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link)
External links
[edit]- Phosphofructokinase-1 at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Proteopedia.org Phosphofructokinase