Sulfolene
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
2,5-Dihydro-1H-1λ6-thiophene-1,1-dione | |||
| Other names
2,5-Dihydrothiophene 1,1-dioxide
Butadiene sulfone 3-Sulfolene | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.000.964 | ||
| EC Number |
| ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C4H6O2S | |||
| Molar mass | 118.15 g·mol−1 | ||
| Melting point | 65 to 66 °C (149 to 151 °F; 338 to 339 K) | ||
| Hazards | |||
| GHS labelling:[2] | |||
 
| |||
| Danger | |||
| H318 | |||
| P264+P265, P280, P305+P351+P338, P305+P354+P338, P317, P337+P317 | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Sulfolene, or butadiene sulfone is a cyclic organic chemical with a sulfone functional group. It is a white, odorless, crystalline, indefinitely storable solid, which dissolves in water and many organic solvents.[3] The compound is used as a source of butadiene.[4]
Production
[edit]
Sulfolene is formed by the cheletropic reaction between butadiene and sulfur dioxide. The reaction is typically conducted in an autoclave. Small amounts of hydroquinone or pyrogallol are added to inhibit polymerization of the diene. The reaction proceeds at room temperature over the course of days. At 130 °C, only 30 minutes are required.[5] An analogous procedure gives the isoprene-derived sulfone.[6]
Reactions
[edit]Acid-base reactivity
[edit]The compound is unaffected by acids. It can even be recrystallized from conc. HNO3.[7][8]
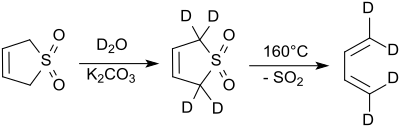
The protons in the 2- and 5-positions rapidly exchange with deuterium oxide under alkaline conditions.[9] Sodium cyanide catalyzes this reaction.[10]
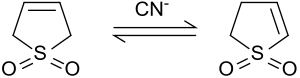
Isomerization to 2-sulfolene
[edit]In the presence of base or cyanide, 3-sulfolene isomerizes to a mixture of 2-sulfolene and 3-sulfolene.[10]
At 50 °C an equilibrium mixture is obtained containing 42% 3-sulfolene and 58% 2-sulfolene.[11] The thermodynamically more stable 2-sulfolene can be isolated from the mixture of isomers as pure substance in the form of white plates (m.p. 48-49 °C) by heating for several days at 100 °C, because of the thermal decomposition of the 3-sulfolene at temperatures above 80 °C.[12]
Hydrogenation
[edit]Catalytic hydrogenation yields sulfolane, a solvent used in the petrochemical industry for the extraction of aromatics from hydrocarbon streams. The hydrogenation of 3-sulfolene over Raney nickel at approx. 20 bar and 60 °C gives sulfolane in yields of up to 65% only because of the poisoning of the catalyst by sulfur compounds.[13]
Halogenation
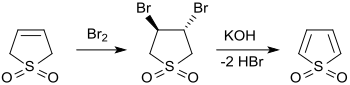
[edit]3-Sulfolene reacts in aqueous solution with bromine to give 3,4-dibromotetrohydrothiophene-1,1-dioxide, which can be dehydrobrominated to thiophene-1,1-dioxide with silver carbonate.[7]
Thiophene-1,1-dioxide, a highly reactive species, is also accessible via the formation of 3,4-bis(dimethylamino)tetrahydrothiophene-1,1-dioxide and successive double quaternization with methyl iodide and Hofmann elimination with silver hydroxide.[12] A less cumbersome two-step synthesis is the two-fold dehydrobromination of 3,4-dibromotetrohydrothiophene-1,1-dioxide with either powdered sodium hydroxide in tetrahydrofuran (THF)[14] or with ultrasonically dispersed metallic potassium.[15]
Diels-Alder reactions
[edit]3-sulfolene is mainly valued as a stand-in for butadiene.[3][4] The in situ production and immediate consumption of 1,3-butadiene largely avoids contact with the diene, which is a gas at room temperature. One potential drawback, aside from expense, is that the evolved sulfur dioxide can cause side reactions with acid-sensitive substrates.
Diels-Alder reaction between 1,3-butadiene and dienophiles of low reactivity usually requires prolonged heating above 100 °C. Such procedures are rather dangerous. If neat butadiene is used, special equipment for work under elevated pressure is required. With sulfolene no buildup of butadiene pressure could be expected as the liberated diene is consumed in the cycloaddition, and therefore the equilibrium of the reversible extrusion reaction acts as an internal "safety valve".[16]
3-Sulfolene reacts with maleic anhydride in boiling xylene to cis-4-cyclohexene-1,2-dicarboxylic anhydride, obtaining yields of up to 90%.[4]
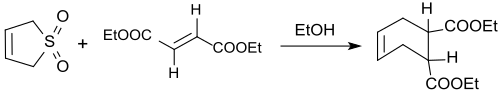
3-Sulfolene reacts also with dienophiles in trans configuration (such as diethyl fumarate) at 110 °C with SO2 elimination in 66–73% yield to the trans-4-cyclohexene-1,2-dicarboxylic diethyl ester.[17]
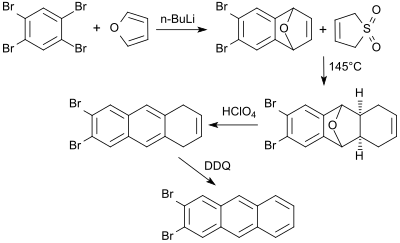
6,7-Dibromo-1,4-epoxy-1,4-dihydronaphthalene (6,7-Dibromonaphthalene-1,4-endoxide, accessible after debromination from 1,2,4,5-tetrabromobenzene using an equivalent of n-butyllithium and Diels-Alder reaction in furan in 70% yield[18]) reacts with 3-sulfolene in boiling xylene to give a tricyclic adduct. This precursor yields, after treatment with perchloric acid, a dibromo dihydroanthracene which is dehydrogenated in the last step with 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) to 2,3-dibromoanthracene.[19]
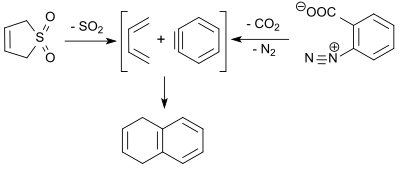
1,3-Butadiene (formed in the retro-cheletrophic reaction of 3-sulfolene) reacts with dehydrobenzene (benzyne, obtained by thermal decomposition of benzenediazonium-2-carboxylate) in a Diels-Alder reaction in 9% yield to give 1,4-dihydronaphthalene.[20]
2- and 3-Sulfolenes as a dienophile
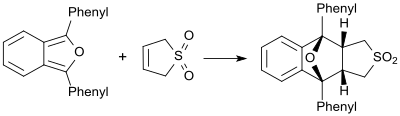
[edit]In the presence of very reactive dienes (for example 1,3-diphenylisobenzofuran) butadienesulfone behaves as a dienophile and forms the corresponding Diels-Alder adduct.[21]
As early as 1938, Kurt Alder and co-workers reported Diels-Alder adducts from the isomeric 2-sulfolene with 1,3-butadiene and 2-sulfolene with cyclopentadiene.[22]
Other cycloadditions
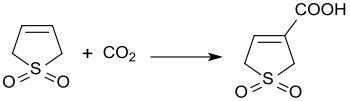
[edit]The base-catalyzed reaction of 3-sulfolene with carbon dioxide at 3 bar pressure produces 3-sulfolene-3-carboxylic acid in 45% yield.[23]
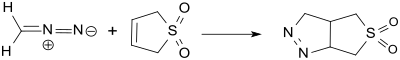
With diazomethane, 3-sulfolene forms in a 1,3-dipolar cycloadduct:[24]
Polymerization
[edit]In 1935, H. Staudinger and co-workers found that the reaction of butadiene and SO2 at room temperature gives a second product in addition to 3-sulfolene. This second product is an amorphous solid polymer. By free-radical polymerization of 3-sulfolene in peroxide-containing diethyl ether, up to 50% insoluble high-molecular-weight poly-sulfolene was obtained. The polymer resists degradation by sulfuric and nitric acids.[8]
In subsequent investigations, polymerization of 3-sulfolene was initiated above 100 °C with the radical initiator azobis(isobutyronitrile) (AIBN).[25] 3-sulfolene does not copolymerize with vinyl compounds, however. On the other hand, 2-sulfolene does not homopolymerize, but forms copolymers with vinyl compounds, e.g. acrylonitrile and vinyl acetate.
3-Sulfolene as a recyclable solvent
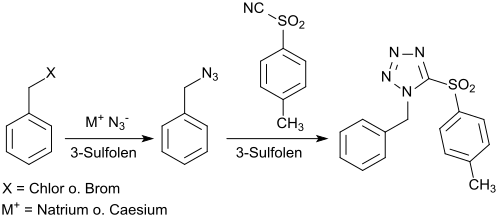
[edit]The reversibility of the interconversion of 3-sulfolene with buta-1,3-diene and sulfur dioxide suggests the use of sulfolene as a recyclable aprotic dipolar solvent, in replacement for dimethyl sulfoxide (DMSO), which is often used but difficult to separate and poorly reusable.[26] As a model reaction, the reaction of benzyl azide with 4-toluenesulfonic acid cyanide forming 1-benzyl-5-(4-toluenesulfonyl)tetrazole was investigated. The formation of the tetrazole can also be carried out as a one-pot reaction without the isolation of the benzyl azide with 72% overall yield.
After the reaction, the solvent 3-sulfolene is decomposed at 135 °C and the volatile butadiene (b.p. −4.4 °C) and sulfur dioxide (b.p. −10.1 °C) are deposited in a cooling trap at −76 °C charged with excess sulfur dioxide. After the addition of hydroquinone as polymerization inhibition, 3-sulfoles is formed again quantitatively upon heating to room temperature. It appears questionable though, if 3-sulfolene with a useful liquid phase range of only 64 to a maximum of about 100 °C can be used as DMSO substitutes (easy handling, low cost, environmental compatibility) in industrial practice.
Uses
[edit]Aside from its synthetic versatility (see above), sulfolene is used as an additive in electrochemical fluorination. It can increase the yield of perfluorooctanesulfonyl fluoride by about 70%.[27] It is "highly soluble in anhydrous HF and increases the conductivity of the electrolyte solution".[27] In this application, it undergoes a ring opening and is fluorinated to form perfluorobutanesulfonyl fluoride.
Further reading
[edit]- DE 506839, H. Staudinger, "Verfahren zur Darstellung von monomolekularen Reaktionsprodukten von ungesättigten Kohlenwasserstoffen der Butadienreihe mit Schwefeldioxyd", published 1930-08-28, assigned to H. Staudinger
References
[edit]- ^ Sulfolene at Sigma-Aldrich
- ^ "3-Sulfolene". pubchem.ncbi.nlm.nih.gov.
- ^ a b J. M. McIntosh (2001). "3-Sulfolene". E-EROS Encyclopedia of Reagents for Organic Synthesis. doi:10.1002/047084289X.rs130. ISBN 0471936235.
- ^ a b c Sample, Thomas E.; Hatch, Lewis F. (Jan 1968). "3-Sulfolene: A Butadiene Source for a Diels-Alder Synthesis". Journal of Chemical Education. 45 (1): 55. doi:10.1021/ed045p55.
- ^ Houben-Weyl (1955). Volume IX: Sulfur, Selenium, Tellurium Compounds. Methods of Organic Chemistry (4th ed.). Stuttgart: Georg Thieme Verlag. p. 237. ISBN 978-3-13-208104-8.
- ^ Robert L. Frank, Raymond P. Seven (1949). "Isoprene Cyclic Sulfone". Organic Syntheses. 29: 59. doi:10.15227/orgsyn.029.0059.
- ^ a b DE 506839, H. Staudinger, "Verfahren zur Darstellung von monomolekularen Reaktionsprodukten von ungesättigten Kohlenwasserstoffen der Butadienreihe mit Schwefeldioxyd", published 1930-8-28, assigned to H. Staudinger
- ^ a b H. Staudinger; B. Ritzenthaler (1935). "Über hochmolekulare Verbindungen, 104. Mitteil.: Über die Anlagerung von Schwefeldioxyd an Äthylen-Derivate". Chemische Berichte (in German). 68 (3): 455–471. doi:10.1002/cber.19350680317.
- ^ J. Leonard; A. B. Hague; J. A. Knight (1998). "6. Preparation of substituted 3-sulfolenes and their use as precursors for Diels-Alder dienes". Organosulfur Chemistry. Vol. 2 (4th ed.). San Diego: Academic Press, Inc. p. 241. ISBN 0-12-543562-2.
- ^ a b US 4187231, R. L. Cobb, "Cyanide-catalyzed isomerization and deuterium exchange with alpha- and beta-sulfolenes", published 1980-02-05, assigned to Phillips Petroleum Co.
- ^ C. D. Broaddus (1968). "Equilibria and base-catalyzed exchange of substituted olefins". Accounts of Chemical Research. 1 (8): 231–238. doi:10.1021/ar50008a002.
- ^ a b W. J. Bailey; E. W. Cummins (1954). "Cyclic dienes. III. The synthesis of thiophene-1,1-dioxide". Journal of the American Chemical Society. 76 (7): 1932–1936. doi:10.1021/ja01636a058.
- ^ US 4286099, M. E. Nash, E. E. Huxley, "Sulfolene hydrogenation", published 1981-08-25, assigned to Phillips Petroleum Co.
- ^ D. M. Lemal; G. D. Goldman (1988). "Synthesis of azulene, a blue hydrocarbon". Journal of Chemical Education. 65 (10): 923. Bibcode:1988JChEd..65..923L. doi:10.1021/ed065p923.
- ^ T.-S. Chou; M.-M. Chen (1987). "Chemoselective reactions of ultrasonically dispersed potassium with some brominated hydrothiophene-S,S-dioxides". Heterocycles. 26 (11): 2829–2834. doi:10.3987/R-1987-11-2829.
- ^ M. A. Filatov; S. Baluschev; I. Z. Ilieva; V. Enkelmann; T. Miteva; K. Landfester; S. E. Aleshchenkov; A. V. Cheprakov (2012). "Tetraaryltetraanthra[2,3]porphyrins: Synthesis, Structure, and Optical Properties". The Journal of Organic Chemistry. 77 (24): 11119–11131. doi:10.1021/jo302135q. PMID 23205621.
- ^ "Diethyl trans-Δ4-tetrahydrophthalate". Organic Syntheses. 50. doi:10.15227/orgsyn.050.0043.
- ^ H. Hart, A. Bashir-Hashemi, J. Luo, M. A. Meador (1986). "Iptycenes: Extended Triptycenes". Tetrahedron. 42: 1641–1654. doi:10.1016/S0040-4020(01)87581-5.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ C.-T. Lin, T.-C. Chou (1988). "Synthesis of 2,3-dibromoanthracene". Synthesis. 1988 (8): 628–630. doi:10.1055/s-1988-27659. S2CID 93109532.
- ^ L. F. Hatch, D. Peter (1968). "Reaction of benzyne with butadiene". Chemical Communications. 23 (23): 1499. doi:10.1039/C19680001499.
- ^ M. P. Cava, J. P. VanMeter (1969). "Condensed cyclobutane aromatic compounds. XXX. Synthesis of some unusual 2,3-naphthoquinonoid heterocycles. A synthetic route to derivatives of naphtho[2,3-b]biphenylene and anthra[b]cyclobutene". The Journal of Organic Chemistry. 34 (3): 538–545. doi:10.1021/jo01255a012.
- ^ K. Alder; H. F. Rickert; E. Windemuth (1938). "Zur Kenntnis der Dien-Synthese, X. Mitteil.: Über die Dien-Synthese mit α, β-ungesättigten Nitrokörpern, Sulfonen und Thio-Äthern". Chemische Berichte. 71 (12): 2451–2461. doi:10.1002/cber.19380711206.
- ^ G. S. Andrade; et al. (2003). "The one-pot synthesis and Diels-Alder Reactivity of 2,5-dihydrothiophene-1,1-dioxide-3-carboxylic acid". Synthetic Communications. 33 (20): 3643–3650. doi:10.1081/SCC-120024845. S2CID 98504228.
- ^ M. E. Brant; J. E. Wulff (2016). "3-Sulfolenes and their derivatives: Synthesis and applications". Synthesis. 48 (1): 1–17. doi:10.1055/s-0035-1560351. S2CID 196826278.
- ^ E. J. Goethals (1967). "On the polymerization and copolymerization of sulfolenes". Macromolecular Chemistry and Physics. 109 (1): 132–142. doi:10.1002/macp.1967.021090113.
- ^ Y. Huang; et al. (2015). "Butadiene sulfone as 'volatile', recyclable dipolar, aprotic solvent for conducting substitution and cycloaddition reactions". Sustainable Chemical Processes. 3 (13). doi:10.1186/s40508-015-0040-7.
- ^ a b Lehmler HJ (March 2005). "Synthesis of environmentally relevant fluorinated surfactants—a review". Chemosphere. 58 (11): 1471–96. Bibcode:2005Chmsp..58.1471L. doi:10.1016/j.chemosphere.2004.11.078. PMID 15694468.