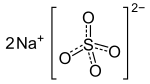
Sodium sulfate
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Sodium sulfate
| |||
| Other names
Sodium sulphate
Disodium sulfate Sulfate of sodium Thénardite (anhydrous mineral) Glauber's salt (decahydrate) Sal mirabilis (decahydrate) Mirabilite (decahydrate mineral) | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.028.928 | ||
| E number | E514(i) (acidity regulators, ...) | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII |
| ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| Na2SO4 | |||
| Molar mass | 142.04 g/mol (anhydrous) 322.20 g/mol (decahydrate) | ||
| Appearance | white crystalline solid hygroscopic | ||
| Odor | odorless | ||
| Density | 2.664 g/cm3 (anhydrous) 1.464 g/cm3 (decahydrate) | ||
| Melting point | 884 °C (1,623 °F; 1,157 K) (anhydrous) 32.38 °C (decahydrate) | ||
| Boiling point | 1,429 °C (2,604 °F; 1,702 K) (anhydrous) | ||
| anhydrous: 4.76 g/100 mL (0 °C) 28.1 g/100 mL (25 °C)[1] 42.7 g/100 mL (100 °C) heptahydrate: 19.5 g/100 mL (0 °C) 44 g/100 mL (20 °C) | |||
| Solubility | insoluble in ethanol soluble in glycerol, water, and hydrogen iodide | ||
| −52.0·10−6 cm3/mol | |||
Refractive index (nD)
|
1.468 (anhydrous) 1.394 (decahydrate) | ||
| Structure | |||
| orthorhombic (anhydrous)[2] monoclinic (decahydrate) | |||
| Pharmacology | |||
| A06AD13 (WHO) A12CA02 (WHO) | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Irritant | ||
| NFPA 704 (fire diamond) | |||
| Flash point | Non-flammable | ||
| Safety data sheet (SDS) | ICSC 0952 | ||
| Related compounds | |||
Other anions
|
Sodium selenate Sodium tellurate | ||
Other cations
|
Lithium sulfate Potassium sulfate Rubidium sulfate Caesium sulfate | ||
Related compounds
|
Sodium bisulfate Sodium sulfite Sodium persulfate Sodium pyrosulfate | ||
| Supplementary data page | |||
"ARMADURA Z29 HELMET ARMOR Z29" by OSCAR CREATIVO | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Sodium sulfate (also known as sodium sulphate or sulfate of soda) is the inorganic compound with formula Na2SO4 as well as several related hydrates. All forms are white solids that are highly soluble in water. With an annual production of 6 million tonnes, the decahydrate is a major commodity chemical product. It is mainly used as a filler in the manufacture of powdered home laundry detergents and in the Kraft process of paper pulping for making highly alkaline sulfides.[3]
Forms
[edit]- Anhydrous sodium sulfate, known as the rare mineral thenardite, used as a drying agent in organic synthesis.
- Heptahydrate sodium sulfate, a very rare form.
- Decahydrate sodium sulfate, known as the mineral mirabilite, widely used by chemical industry. It is also known as Glauber's salt.
History
[edit]The decahydrate of sodium sulfate is known as Glauber's salt after the Dutch–German chemist and apothecary Johann Rudolf Glauber (1604–1670), who discovered it in Austrian spring water in 1625. He named it sal mirabilis (miraculous salt), because of its medicinal properties: the crystals were used as a general-purpose laxative, until more sophisticated alternatives came about in the 1900s.[4][5] However, J. Kunckel later alleged that it was known as a secret medicine in Saxony already in the mid-16th century.[6]
In the 18th century, Glauber's salt began to be used as a raw material for the industrial production of soda ash (sodium carbonate), by reaction with potash (potassium carbonate). Demand for soda ash increased, and the supply of sodium sulfate had to increase in line. Therefore, in the 19th century, the large-scale Leblanc process, producing synthetic sodium sulfate as a key intermediate, became the principal method of soda-ash production.[7]
Chemical properties
[edit]Sodium sulfate is a typical electrostatically bonded ionic sulfate. The existence of free sulfate ions in solution is indicated by the easy formation of insoluble sulfates when these solutions are treated with Ba2+ or Pb2+ salts:
- Na2SO4 + BaCl2 → 2 NaCl + BaSO4
Sodium sulfate is unreactive toward most oxidizing or reducing agents. At high temperatures, it can be converted to sodium sulfide by carbothermal reduction (aka thermo-chemical sulfate reduction (TSR), high temperature heating with charcoal, etc.):[8]
- Na2SO4 + 2 C → Na2S + 2 CO2
This reaction was employed in the Leblanc process, a defunct industrial route to sodium carbonate.
Sodium sulfate reacts with sulfuric acid to give the acid salt sodium bisulfate:[9][10]
- Na2SO4 + H2SO4 ⇌ 2 NaHSO4
Sodium sulfate displays a moderate tendency to form double salts. The only alums formed with common trivalent metals are NaAl(SO4)2 (unstable above 39 °C) and NaCr(SO4)2, in contrast to potassium sulfate and ammonium sulfate which form many stable alums.[11] Double salts with some other alkali metal sulfates are known, including Na2SO4·3K2SO4 which occurs naturally as the mineral aphthitalite. Formation of glaserite by reaction of sodium sulfate with potassium chloride has been used as the basis of a method for producing potassium sulfate, a fertiliser.[12] Other double salts include 3Na2SO4·CaSO4, 3Na2SO4·MgSO4 (vanthoffite) and NaF·Na2SO4.[13]
Physical properties
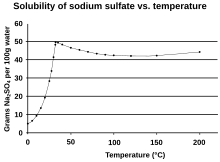
[edit]Sodium sulfate has unusual solubility characteristics in water.[14] Its solubility in water rises more than tenfold between 0 °C and 32.384 °C, where it reaches a maximum of 49.7 g/100 mL. At this point the solubility curve changes slope, and the solubility becomes almost independent of temperature. This temperature of 32.384 °C, corresponding to the release of crystal water and melting of the hydrated salt, serves as an accurate temperature reference for thermometer calibration.
Structure
[edit]Crystals of the decahydrate consist of [Na(OH2)6]+ ions with octahedral molecular geometry. These octahedra share edges such that 8 of the 10 water molecules are bound to sodium and 2 others are interstitial, being hydrogen-bonded to sulfate. These cations are linked to the sulfate anions by hydrogen bonds. The Na–O distances are about 240 pm.[15] Crystalline sodium sulfate decahydrate is also unusual among hydrated salts in having a measurable residual entropy (entropy at absolute zero) of 6.32 J/(K·mol). This is ascribed to its ability to distribute water much more rapidly compared to most hydrates.[16]
Production
[edit]The world production of sodium sulfate, almost exclusively in the form of the decahydrate, amounts to approximately 5.5 to 6 million tonnes annually (Mt/a). In 1985, production was 4.5 Mt/a, half from natural sources, and half from chemical production. After 2000, at a stable level until 2006, natural production had increased to 4 Mt/a, and chemical production decreased to 1.5 to 2 Mt/a, with a total of 5.5 to 6 Mt/a.[17][18][19][20] For all applications, naturally produced and chemically produced sodium sulfate are practically interchangeable.
Natural sources
[edit]Two thirds of the world's production of the decahydrate (Glauber's salt) is from the natural mineral form mirabilite, for example as found in lake beds in southern Saskatchewan. In 1990, Mexico and Spain were the world's main producers of natural sodium sulfate (each around 500,000 tonnes), with Russia, United States, and Canada around 350,000 tonnes each.[18] Natural resources are estimated at over 1 billion tonnes.[17][18]
Major producers of 200,000 to 1,500,000 tonnes/year in 2006 included Searles Valley Minerals (California, US), Airborne Industrial Minerals (Saskatchewan, Canada), Química del Rey (Coahuila, Mexico), Minera de Santa Marta and Criaderos Minerales Y Derivados, also known as Grupo Crimidesa (Burgos, Spain), Minera de Santa Marta (Toledo, Spain), Sulquisa (Madrid, Spain), Chengdu Sanlian Tianquan Chemical (Tianquan County, Sichuan, China), Hongze Yinzhu Chemical Group (Hongze District, Jiangsu, China), Nafine Chemical Industry Group (Shanxi, China), Sichuan Province Chuanmei Mirabilite (万胜镇, Dongpo District, Meishan, Sichuan, China), and Kuchuksulphat JSC (Altai Krai, Siberia, Russia).[17][19]
Anhydrous sodium sulfate occurs in arid environments as the mineral thenardite. It slowly turns to mirabilite in damp air. Sodium sulfate is also found as glauberite, a calcium sodium sulfate mineral. Both minerals are less common than mirabilite.[citation needed]
Chemical industry
[edit]About one third of the world's sodium sulfate is produced as by-product of other processes in chemical industry. Most of this production is chemically inherent to the primary process, and only marginally economical. By effort of the industry, therefore, sodium sulfate production as by-product is declining.
The most important chemical sodium sulfate production is during hydrochloric acid production, either from sodium chloride (salt) and sulfuric acid, in the Mannheim process, or from sulfur dioxide in the Hargreaves process.[21] The resulting sodium sulfate from these processes is known as salt cake.
- Mannheim: 2 NaCl + H2SO4 → 2 HCl + Na2SO4
- Hargreaves: 4 NaCl + 2 SO2 + O2 + 2 H2O → 4 HCl + 2 Na2SO4
The second major production of sodium sulfate are the processes where surplus sodium hydroxide is neutralised by sulfuric acid to obtain sulfate (SO2−4) by using copper sulfate (CuSO4) (as historically applied on a large scale in the production of rayon by using copper(II) hydroxide). This method is also a regularly applied and convenient laboratory preparation.
- 2 NaOH(aq) + H2SO4(aq) → Na2SO4(aq) + 2 H2O(l) ΔH = -112.5 kJ (highly exothermic)
In the laboratory it can also be synthesized from the reaction between sodium bicarbonate and magnesium sulfate, by precipitating magnesium carbonate.
- 2 NaHCO3 + MgSO4 → Na2SO4 + MgCO3 + CO2 + H2O
However, as commercial sources are readily available, laboratory synthesis is not practised often. Formerly, sodium sulfate was also a by-product of the manufacture of sodium dichromate, where sulfuric acid is added to sodium chromate solution forming sodium dichromate, or subsequently chromic acid. Alternatively, sodium sulfate is or was formed in the production of lithium carbonate, chelating agents, resorcinol, ascorbic acid, silica pigments, nitric acid, and phenol.[17]
Bulk sodium sulfate is usually purified via the decahydrate form, since the anhydrous form tends to attract iron compounds and organic compounds. The anhydrous form is easily produced from the hydrated form by gentle warming.
Major sodium sulfate by-product producers of 50–80 Mt/a in 2006 include Elementis Chromium (chromium industry, Castle Hayne, NC, US), Lenzing AG (200 Mt/a, rayon industry, Lenzing, Austria), Addiseo (formerly Rhodia, methionine industry, Les Roches-Roussillon, France), Elementis (chromium industry, Stockton-on-Tees, UK), Shikoku Chemicals (Tokushima, Japan) and Visko-R (rayon industry, Russia).[17]
Applications
[edit]Commodity industries
[edit]With US pricing at $30 per tonne in 1970, up to $90 per tonne for salt cake quality, and $130 for better grades, sodium sulphate is a very cheap material. The largest use is as filler in powdered home laundry detergents, consuming approximately 50% of world production. This use is waning as domestic consumers are increasingly switching to compact or liquid detergents that do not include sodium sulfate.[17]
Papermaking
[edit]Another formerly major use for sodium sulfate, notably in the US and Canada, is in the Kraft process for the manufacture of wood pulp. Organics present in the "black liquor" from this process are burnt to produce heat, needed to drive the reduction of sodium sulfate to sodium sulfide. However, due to advances in the thermal efficiency of the Kraft recovery process in the early 1960s, more efficient sulfur recovery was achieved and the need for sodium sulfate makeup was drastically reduced.[22] Hence, the use of sodium sulfate in the US and Canadian pulp industry declined from 1,400,000 tonnes per year in 1970 to only approx. 150,000 tonnes in 2006.[17]
Glassmaking
[edit]The glass industry provides another significant application for sodium sulfate, as second largest application in Europe. Sodium sulfate is used as a fining agent, to help remove small air bubbles from molten glass. It fluxes the glass, and prevents scum formation of the glass melt during refining. The glass industry in Europe has been consuming from 1970 to 2006 a stable 110,000 tonnes annually.[17]
Textiles
[edit]Sodium sulfate is important in the manufacture of textiles, particularly in Japan, where this is the largest application. Sodium sulfate is added to increase the ionic strength of the solution and so helps in "levelling", i.e. reducing negative electrical charges on textile fibres, so that dyes can penetrate evenly (see the theory of the diffuse double layer (DDL) elaborated by Gouy and Chapman). Unlike the alternative sodium chloride, it does not corrode the stainless steel vessels used in dyeing. This application in Japan and US consumed in 2006 approximately 100,000 tonnes.[17]
Food industry
[edit]Sodium sulfate is used as a diluent for food colours.[23] It is known as E number additive E514.
Heat storage
[edit]The high heat-storage capacity in the phase change from solid to liquid, and the advantageous phase change temperature of 32 °C (90 °F) makes this material especially appropriate for storing low-grade solar heat for later release in space heating applications. In some applications the material is incorporated into thermal tiles that are placed in an attic space, while in other applications, the salt is incorporated into cells surrounded by solar–heated water. The phase change allows a substantial reduction in the mass of the material required for effective heat storage (the heat of fusion of sodium sulfate decahydrate is 82 kJ/mol or 252 kJ/kg[24]), with the further advantage of a consistency of temperature as long as sufficient material in the appropriate phase is available.
For cooling applications, a mixture with common sodium chloride salt (NaCl) lowers the melting point to 18 °C (64 °F). The heat of fusion of NaCl·Na2SO4·10H2O, is actually increased slightly to 286 kJ/kg.[25]
Small-scale applications
[edit]In the laboratory, anhydrous sodium sulfate is widely used as an inert drying agent, for removing traces of water from organic solutions.[26] It is more efficient, but slower-acting, than the similar agent magnesium sulfate. It is only effective below about 30 °C (86 °F), but it can be used with a variety of materials since it is chemically fairly inert. Sodium sulfate is added to the solution until the crystals no longer clump together; the two video clips (see above) demonstrate how the crystals clump when still wet, but some crystals flow freely once a sample is dry.
Glauber's salt, the decahydrate, is used as a laxative. It is effective for the removal of certain drugs, such as paracetamol (acetaminophen) from the body; thus it can be used after an overdose.[27][28]
In 1953, sodium sulfate was proposed for heat storage in passive solar heating systems. This takes advantage of its unusual solubility properties, and the high heat of crystallisation (78.2 kJ/mol).[29]
Other uses for sodium sulfate include de-frosting windows, starch manufacture, as an additive in carpet fresheners, and as an additive to cattle feed.
At least one company, Thermaltake, makes a laptop computer chill mat (iXoft Notebook Cooler) using sodium sulfate decahydrate inside a quilted plastic pad. The material slowly turns to liquid and recirculates, equalizing laptop temperature and acting as an insulation.[30]
Safety
[edit]Although sodium sulfate is generally regarded as non-toxic,[23] it should be handled with care. The dust can cause temporary asthma or eye irritation; this risk can be prevented by using eye protection and a paper mask. Transport is not limited, and no Risk Phrase or Safety Phrase applies.[31]
References
[edit]- ^ National Center for Biotechnology Information. PubChem Compound Summary for CID 24436, Sodium sulfate. https://pubchem.ncbi.nlm.nih.gov/compound/Sodium-sulfate. Accessed Nov. 2, 2020.
- ^ Zachariasen WH, Ziegler GE (1932). "The crystal structure of anhydrous sodium sulfate Na2SO4". Zeitschrift für Kristallographie, Kristallgeometrie, Kristallphysik, Kristallchemie. 81 (1–6). Wiesbaden: Akademische Verlagsgesellschaft: 92–101. doi:10.1524/zkri.1932.81.1.92. S2CID 102107891.
- ^ Helmold Plessen (2000). "Sodium Sulfates". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a24_355. ISBN 978-3527306732.
- ^ Szydlo, Zbigniew (1994). Water which does not wet hands: The Alchemy of Michael Sendivogius. London–Warsaw: Polish Academy of Sciences.
- ^ Westfall, Richard S. (1995). "Glauber, Johann Rudolf". The Galileo Project. Archived from the original on 2011-11-18.
- ^ Chisholm, Hugh, ed. (1911). "Glauber's Salt". Encyclopædia Britannica (11th ed.). Cambridge University Press.
- ^ Aftalion, Fred (1991). A History of the International Chemical Industry. Philadelphia: University of Pennsylvania Press. pp. 11–16. ISBN 978-0-8122-1297-6.
- ^ Handbook of Chemistry and Physics (71st ed.). Ann Arbor, Michigan: CRC Press. 1990. ISBN 9780849304712.
- ^ The Merck Index (7th ed.). Rahway, New Jersey, US: Merck & Co. 1960.
- ^ Nechamkin, Howard (1968). The Chemistry of the Elements. New York: McGraw-Hill.
- ^ Lipson, Henry; Beevers, C. A. (1935). "The Crystal Structure of the Alums". Proceedings of the Royal Society A. 148 (865): 664–80. Bibcode:1935RSPSA.148..664L. doi:10.1098/rspa.1935.0040.
- ^ Garrett, Donald E. (2001). Sodium sulfate: handbook of deposits, processing, properties, and use. San Diego: Academic Press. ISBN 978-0-12-276151-5.
- ^ Mellor, Joseph William (1961). Mellor's Comprehensive Treatise on Inorganic and Theoretical Chemistry. Vol. II (new impression ed.). London: Longmans. pp. 656–673. ISBN 978-0-582-46277-9.
- ^ Linke, W. F.; A. Seidell (1965). Solubilities of Inorganic and Metal Organic Compounds (4th ed.). Van Nostrand. ISBN 978-0-8412-0097-5.
- ^ Helena W. Ruben, David H. Templeton, Robert D. Rosenstein, Ivar Olovsson, "Crystal Structure and Entropy of Sodium Sulfate Decahydrate", J. Am. Chem. Soc. 1961, volume 83, pp. 820–824. doi:10.1021/ja01465a019.
- ^ Brodale, G.; W. F. Giauque (1958). "The Heat of Hydration of Sodium Sulfate. Low Temperature Heat Capacity and Entropy of Sodium Sulfate Decahydrate". Journal of the American Chemical Society. 80 (9): 2042–2044. doi:10.1021/ja01542a003.
- ^ a b c d e f g h i Suresh, Bala; Kazuteru Yokose (May 2006). Sodium sulfate. Zurich: Chemical Economic Handbook SRI Consulting. pp. 771.1000A–771.1002J. Archived from the original on 2007-03-14.
{{cite book}}:|work=ignored (help) - ^ a b c "Statistical compendium Sodium sulfate". Reston, Virginia: US Geological Survey, Minerals Information. 1997. Archived from the original on 2007-03-07. Retrieved 2007-04-22.
- ^ a b The economics of sodium sulphate (Eighth ed.). London: Roskill Information Services. 1999.
- ^ The sodium sulphate business. London: Chem Systems International. November 1984.
- ^ Butts, D. (1997). Kirk-Othmer Encyclopedia of Chemical Technology. Vol. v22 (4th ed.). pp. 403–411.
- ^ Smook, Gary (2002). Handbook for Pulp and Paper Technologists. p. 143. Archived from the original on 2016-08-07.
- ^ a b "Sodium sulfate (WHO Food Additives Series 44)". World Health Organization. 2000. Archived from the original on 2007-09-04. Retrieved 2007-06-06.
- ^ "Phase-Change Materials for Low-Temperature Solar Thermal Applications" (PDF). Archived (PDF) from the original on 2015-09-24. Retrieved 2014-06-19.
- ^ "Phase-Change Materials for Low-Temperature Solar Thermal Applications" (PDF). p. 8. Archived (PDF) from the original on 2015-09-24. Retrieved 2014-06-19.
- ^ Vogel, Arthur I.; B.V. Smith; N.M. Waldron (1980). Vogel's Elementary Practical Organic Chemistry 1 Preparations (3rd ed.). London: Longman Scientific & Technical.
- ^ Cocchetto, D.M.; G. Levy (1981). "Absorption of orally administered sodium sulfate in humans". J Pharm Sci. 70 (3): 331–3. doi:10.1002/jps.2600700330. PMID 7264905.
- ^ Prescott, L. F.; Critchley, J. A. J. H. (1979). "The Treatment of Acetaminophen Poisoning". Annual Review of Pharmacology and Toxicology. 23: 87–101. doi:10.1146/annurev.pa.23.040183.000511. PMID 6347057.
- ^ Telkes, Maria (1953). Improvements in or relating to a device and a composition of matter for the storage of heat.
{{cite book}}:|work=ignored (help) - ^ "IXoft Specification". Thermaltake Technology Co., Ltd. Archived from the original on 2016-03-12. Retrieved 2015-08-15.
- ^ "MSDS Sodium Sulfate Anhydrous". James T Baker. 2006. Archived from the original on 2003-06-19. Retrieved 2007-04-21.
External links
[edit]- Calculators: surface tensions, and densities, molarities, and molalities of aqueous sodium sulfate