Pipemidic acid
Appearance
This article needs additional citations for verification. (August 2014) |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.052.283 |
| Chemical and physical data | |
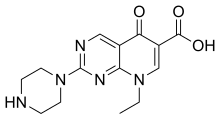
| Formula | C14H17N5O3 |
| Molar mass | 303.322 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Pipemidic acid is a member of the pyridopyrimidine class of antibacterials, which display some overlap in mechanism of action with analogous pyridone-containing quinolones. It was introduced in 1979 and is active against gram negative and some gram positive bacteria. It was used for gastrointestinal, biliary, and urinary infections.[1] The marketing authorization of pipemidic acid has been suspended throughout the EU.[2]
References
[edit]- ^ "Pipemidic Acid - MeSH - NCBI". www.ncbi.nlm.nih.gov. Retrieved 2019-01-20.
- ^ "Disabling and potentially permanent side effects lead to suspension or restrictions of quinolone and fluoroquinolone antibiotics". European Medicines Agency. 11 March 2019.
