Mohs scale

The Mohs scale of mineral hardness (/moʊz/) is a qualitative ordinal scale, from 1 to 10, characterizing scratch resistance of minerals through the ability of harder material to scratch softer material.
The scale was introduced in 1812 by the German geologist and mineralogist Friedrich Mohs, in his book "Versuch einer Elementar-Methode zur naturhistorischen Bestimmung und Erkennung der Fossilien";[1][2] it is one of several definitions of hardness in materials science, some of which are more quantitative.[3]
The method of comparing hardness by observing which minerals can scratch others is of great antiquity, having been mentioned by Theophrastus in his treatise On Stones, c. 300 BC, followed by Pliny the Elder in his Naturalis Historia, c. AD 77.[4][5][6] The Mohs scale is useful for identification of minerals in the field, but is not an accurate predictor of how well materials endure in an industrial setting.[7]
Reference minerals
[edit]The Mohs scale of mineral hardness is based on the ability of one natural sample of mineral to scratch another mineral visibly. The samples of matter used by Mohs are all different minerals. Minerals are chemically pure solids found in nature. Rocks are made up of one or more minerals. As the hardest known naturally occurring substance when the scale was designed, diamonds are at the top of the scale. The hardness of a material is measured against the scale by finding the hardest material that the given material can scratch, or the softest material that can scratch the given material. For example, if some material is scratched by apatite but not by fluorite, its hardness on the Mohs scale would be between 4 and 5.[8]
"Scratching" a material for the purposes of the Mohs scale means creating non-elastic dislocations visible to the naked eye. Frequently, materials that are lower on the Mohs scale can create microscopic, non-elastic dislocations on materials that have a higher Mohs number. While these microscopic dislocations are permanent and sometimes detrimental to the harder material's structural integrity, they are not considered "scratches" for the determination of a Mohs scale number.[9]
Each of the ten hardness values in the Mohs scale is represented by a reference mineral, most of which are widespread in rocks.
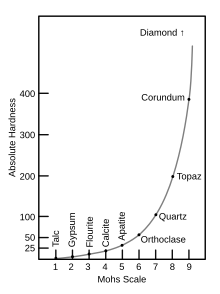
The Mohs scale is an ordinal scale. For example, corundum (9) is twice as hard as topaz (8), but diamond (10) is four times as hard as corundum. The table below shows the comparison with the absolute hardness measured by a sclerometer, with pictorial examples.[10][11]
| Mohs hardness | Reference mineral | Chemical formula | Absolute hardness[12] | Image |
|---|---|---|---|---|
| 1 | Talc | Mg3Si4O10(OH)2 | 1 | 
|
| 2 | Gypsum | CaSO4·2H2O | 2 | 
|
| 3 | Calcite | CaCO3 | 14 | 
|
| 4 | Fluorite | CaF2 | 21 | 
|
| 5 | Apatite | Ca5(PO4)3(OH−,Cl−,F−) | 48 | 
|
| 6 | Orthoclase feldspar | KAlSi3O8 | 72 | 
|
| 7 | Quartz | SiO2 | 100 | 
|
| 8 | Topaz | Al2SiO4(OH−,F−)2 | 200 | 
|
| 9 | Corundum | Al2O3 | 400 | 
|
| 10 | Diamond | C | 1500 | 
|
Examples
[edit]Below is a table of more materials by Mohs scale. Some of them have a hardness between two of the Mohs scale reference minerals. Some solid substances which are not minerals have been assigned a hardness on the Mohs scale. However, if the substance is actually a mixture of other substances, hardness can be difficult to determine or may be misleading or meaningless. For example, some sources have assigned a Mohs hardness of 6 or 7 to granite but it is a rock made of several minerals, each with its own Mohs hardness (e.g. topaz-rich granite contains: topaz - hardness 8, quartz - hardness 7, orthoclase feldspar - hardness 6, plagioclase feldspar - hardness 6 to 6.5, mica - hardness 2 to 4).
This section needs additional citations for verification. (November 2022) |
Use
[edit]Despite its lack of precision, the Mohs scale is relevant for field geologists, who use the scale to roughly identify minerals using scratch kits. The Mohs scale hardness of minerals can be commonly found in reference sheets.
Mohs hardness is useful in milling. It allows assessment of which type of mill and grinding medium will best reduce a given product whose hardness is known.[20]
The scale is used by electronic manufacturers for testing the resilience of flat panel display components (such as cover glass for LCDs or encapsulation for OLEDs), as well as to evaluate the hardness of touch screens in consumer electronics.[21]
Comparison with Vickers scale
[edit]Comparison between Mohs hardness and Vickers hardness:[22]
| Mineral name |
Hardness (Mohs) | Hardness (Vickers) (kg/mm2) |
|---|---|---|
| Tin | 1.5 | VHN10 = 7–9 |
| Bismuth | 2–2.5 | VHN100 = 16–18 |
| Gold | 2.5 | VHN10 = 30–34 |
| Silver | 2.5 | VHN100 = 61–65 |
| Chalcocite | 2.5–3 | VHN100 = 84–87 |
| Copper | 2.5–3 | VHN100 = 77–99 |
| Galena | 2.5 | VHN100 = 79–104 |
| Sphalerite | 3.5–4 | VHN100 = 208–224 |
| Heazlewoodite | 4 | VHN100 = 230–254 |
| Goethite | 5–5.5 | VHN100 = 667 |
| Chromite | 5.5 | VHN100 = 1,278–1,456 |
| Anatase | 5.5–6 | VHN100 = 616–698 |
| Rutile | 6–6.5 | VHN100 = 894–974 |
| Pyrite | 6–6.5 | VHN100 = 1,505–1,520 |
| Bowieite | 7 | VHN100 = 858–1,288 |
| Euclase | 7.5 | VHN100 = 1,310 |
| Chromium | 8.5 | VHN100 = 1,875–2,000 |
See also
[edit]References
[edit]- ^ von Groth, Paul Heinrich (1926). Entwicklungsgeschichte der Mineralogischen Wissenschaften [History of the development of the mineralogical sciences] (in German). Berlin: Springer. p. 250. ISBN 9783662409107.
In demselben Jahre (1812) wurde MOHS als Professor am Joanneum angestellt und veröffentliche den ersten Teil seines Werkes "Versuch einer Elementarmethode zur naturhistorischen Bestimmung und Erkennung der Fossilien", in welcher die bekannte Härteskala aufgestellt wurde.
[In the same year (1812) MOHS was employed as a professor at the Joanneum and published the first part of his work "Attempt at an elementary method for the natural-historical determination and recognition of fossils", in which the well-known hardness scale was set up.] - ^ "Mohs hardness" in Encyclopædia Britannica Online
- ^ "Mohs scale of hardness". Mineralogical Society of America. Retrieved 10 February 2021.
- ^ Theophrastus. Theophrastus on Stones. Retrieved 2011-12-10 – via Farlang.com.
- ^ Pliny the Elder. "Book 37, Chap. 15". Naturalis Historia.
Adamas: Six varieties of it. Two remedies.
- ^ Pliny the Elder. "Book 37, Chap. 76". Naturalis Historia.
The methods of testing precious stones.
- ^ "Hardness". Materials Mechanical Hardness. Non-Destructive Testing Resource Center. Archived from the original on 2014-02-14.
- ^ "Mohs Scale of Mineral Hardness". American Federation of Mineralogical Societies.
- ^ Geels, Kay. "The True Microstructure of Materials", pp. 5–13 in Materialographic Preparation from Sorby to the Present. Struers A/S, Copenhagen, Denmark - archived Mar 7 2016
- ^ Amethyst Galleries' Mineral Gallery What is important about hardness?. galleries.com
- ^ Mineral Hardness and Hardness Scales Archived 2008-10-17 at the Wayback Machine. Inland Lapidary
- ^ Mukherjee, Swapna (2012). Applied Mineralogy: Applications in Industry and Environment. Springer Science & Business Media. p. 373. ISBN 978-94-007-1162-4.
- ^ Samsonov, G.V., ed. (1968). "Mechanical Properties of the Elements". Handbook of the Physicochemical Properties of the Elements. New York: IFI-Plenum. p. 432. doi:10.1007/978-1-4684-6066-7. ISBN 978-1-4684-6068-1.
- ^ a b c "Mohs Hardness Scale: Testing the Resistance to Being Scratched". geology.com. Retrieved 2021-08-09.
- ^ "Mohs Hardness Scale". Fundamental Geologic Principles. National Park Service. Retrieved 18 November 2022.
- ^ a b "Reade Advanced Materials - Mohs' Hardness (Typical) of Abrasives". www.reade.com. Retrieved 2021-08-09.
- ^ "Material hardness tables". www.tedpella.com. Archived from the original on 2015-09-12. Retrieved 2019-05-09.
- ^ "Hardness table" (PDF). Retrieved 2019-05-09.
- ^ Levine, Jonathan B.; Tolbert, Sarah H.; Kaner, Richard B. (2009). "Advancements in the search for superhard ultra-incompressible metal borides" (PDF). Advanced Functional Materials. 19 (22): 3526–3527. doi:10.1002/adfm.200901257. S2CID 98675890. Archived from the original (PDF) on 2016-03-04. Retrieved 2015-12-08.
- ^ "Size reduction, comminution". Grinding and milling. PowderProcess.net. Retrieved 27 October 2017.
- ^ Purdy, Kevin (16 May 2014). "Hardness is not toughness: Why your phone's screen may not scratch, but will shatter". Computerworld. IDG Communications Inc. Retrieved 16 April 2021.
- ^ Ralph, Jolyon. "Welcome to mindat.org". mindat.org. Hudson Institute of Mineralogy. Retrieved April 16, 2017.
Further reading
[edit]- Cordua, William S. (c. 1990). "The Hardness of Minerals and Rocks". Lapidary Digest – via gemcutters.org.
- Raden, Aja (2016). Gem: The Definitive Visual Guide. New York: DK Publishing. ISBN 9781465453563.
