Guanidinopropionic acid
Appearance
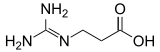
| |
| Names | |
|---|---|
| IUPAC name
3-Carbamimidamidopropanoic acid[1]
| |
| Other names
3-(Diaminomethylideneamino)propanoic acid[citation needed]
| |
| Identifiers | |
3D model (JSmol)
|
|
| 3DMet | |
| 1705262 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.005.937 |
| EC Number |
|
| KEGG | |
| MeSH | guanidopropionic+acid |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H9N3O2 | |
| Molar mass | 131.135 g·mol−1 |
| Appearance | White crystals |
| Odor | Odourless |
| log P | −1.472 |
| Acidity (pKa) | 4.219 |
| Basicity (pKb) | 9.778 |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H315, H319, H335 | |
| P261, P305+P351+P338 | |
| Related compounds | |
Related alkanoic acids
|
|
Related compounds
|
Dimethylacetamide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
β-Guanidinopropionic acid, also referred to as guanidinopropionic acid, beta-guanidinopropionic acid or β-GPA, is a dietary supplement.
β-Guanidinopropionic acid, also known as Ompenaclid (RGX-202), is being investigated in colorectal cancer by Inspirna and Merck[2]
β-Guanidinopropionic acid is a white crystalline powder soluble in water (50 mg/ml-clear, colorless solution).[3]
Studies on animals (rats, monkeys, hamsters) show that acidic guanidine derivatives such as β-GPA can ameliorate hyperglycemia in animal models of noninsulin-dependent diabetes.[4]
Though the oral availability of β-GPA is well established, the basic uptake mechanism has not been studied yet.[5]
References
[edit]- ^ "3-guanidinopropanoic acid (CHEBI:15968)". Chemical Entities of Biological Interest. UK: European Bioinformatics Institute. 20 July 2010. Main. Retrieved 26 April 2012.
- ^ "Pipeline – Inspirna". inspirna.com. Retrieved 2024-11-12.
- ^ Bergeron, R.; Ren, J. M.; Cadman, K. S.; Moore, I. K.; Perret, P.; Pypaert, M.; Young, L. H.; Semenkovich, C. F.; Shulman, G. I. (2001). "Chronic Activation of AMP Kinase Results in NRF-1 Activation and Mitochondrial Biogenesis". American Journal of Physiology. Endocrinology and Metabolism. 281 (6): E1340–E1346. doi:10.1152/ajpendo.2001.281.6.e1340. PMID 11701451. S2CID 21577702.
- ^ Meglasson, M. D.; Wilson, J. M.; Yu, J. H.; Robinson, D. D.; Wyse, B. M.; de Souza, C. J. (September 1993). "Antihyperglycemic Action of Guanidinoalkanoic Acids: 3-Guanidinopropionic Acid Ameliorates Hyperglycemia in Diabetic KKAy and C57BL6Job/ob Mice and Increases Glucose Disappearance in Rhesus Monkeys". The Journal of Pharmacology and Experimental Therapeutics. 266 (3): 1454–1462. PMID 8371149.
- ^ Metzner, L.; Dorn, M.; Markwardt, F.; Brandsch, M. (April 2009). "The Orally Active Antihyperglycemic Drug β-Guanidinopropionic Acid is Transported by the Human Proton-Coupled Amino Acid Transporter hPAT1". Molecular Pharmaceutics. 6 (3): 1006–1011. doi:10.1021/mp9000684. PMID 19358571.
