From Wikipedia, the free encyclopedia
Chemical compound
Pharmaceutical compound
Clomestrone Trade names Arterolo, Atheran, Colesterel, Iposclerone, Liprotene, Persclerol Other names SC-8246; 16α-Chloroestrone 3-methyl ether; 16α-Chloro-3-methoxyestra-1,3,5(10)-trien-17-one Routes of By mouth Drug class Estrogen ; Estrogen ether
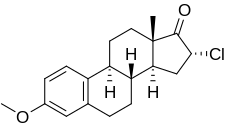
(8R ,9S ,13S ,14S ,16R )-16-chloro-3-methoxy-13-methyl-7,8,9,11,12,14,15,16-octahydro-6H -cyclopenta[a ]phenanthren-17-one
CAS Number PubChem CID ChemSpider UNII CompTox Dashboard (EPA ) ECHA InfoCard 100.021.669 Formula C 19 H 23 Cl O 2 Molar mass −1 3D model (JSmol )
CC12CCC3C(C1CC(C2=O)Cl)CCC4=C3C=CC(=C4)OC
InChI=1S/C19H23ClO2/c1-19-8-7-14-13-6-4-12(22-2)9-11(13)3-5-15(14)16(19)10-17(20)18(19)21/h4,6,9,14-17H,3,5,7-8,10H2,1-2H3/t14-,15-,16+,17-,19+/m1/s1
Key:UQIPVSBPFZSWGD-ILYVXUQDSA-N
Clomestrone (brand names Arterolo , Atheran , Colesterel , Iposclerone , Liprotene , Persclerol , others; former developmental code name SC-8246 ), also known as 16α-chloroestrone 3-methyl ether , is a synthetic , steroidal , weak estrogen derived from estrone and used as an anticholesterolemic agent in the treatment of atherosclerosis .[ 1] [ 2] serum lipid profiles while producing minimal feminization , though some estrogenic side effects , including breast tenderness , loss of libido , and fatigue or avolition , were observed in most patients in clinical studies.[ 3] [ 4] analogue of mytatrienediol , and the two estrogens have similar drug profiles.[ 5] [ 1]
^ a b J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies ISBN 978-1-4757-2085-3 ^ Gregory Pincus (22 October 2013). Hormones and Atherosclerosis: Proceedings of the Conference Held in Brighton, Utah, March 11-14, 1958 ISBN 978-1-4832-7064-7 ^ RIVIN AU (1959). "SC 8246, a new estrogen analog: lipoprotein effects with minimal feminization". Metab. Clin. Exp . 8 : 704–8. PMID 14437693 . SC-8246 (16-alpha chlorestrone 3-methyl ether) was administered for periods of six to twelve months to 20 male survivors of acute myocardial infarction ranging in age from 30 to 63 years. A significant decrease in serum cholesterol concentration occurred in only 6 of 13 patients with an initial cholesterol level above 250 mg. per 100 ml., and there was no change in the other 7. Of 7 initial cholesterol levels below 250 mg. per 100 ml., no level decreased, 3 increased, and 4 were unchanged. In 9 of 11 patients with an initial alpha:beta lipoprotein ratio of less than 20 per cent, a significant increase occurred, but no change in the other 2. Among 9 subjects with a ratio initially above 20 per cent, a further increase occurred in 8 while taking the drug. This estrogen appeared to have an advantage in terms of lessening side-effects. Mild breast tenderness or gynecomastia occurred in 15 of the 17 patients with a "favorable" lipoprotein change. When the dosage was reduced to 5 mg. daily or every other day, the lipoprotein effect in 8 of them could be sustained while the breast changes disappeared. Libido disappeared from 2 patients and was diminished in 1 other. Other side-effects were nausea in 1 patient, loss of ambition in 5, and itching or dryness of the skin in 4. ^ WINSOR T, FISHER EK, PAYNE JH (1959). "A method for the study of peripheral arteriosclerosis". J Am Geriatr Soc . 7 (2): 167–74. doi :10.1111/j.1532-5415.1959.tb01062.x . PMID 13630690 . S2CID 46048052 . ^ Cancer Chemotherapy Abstracts
Estrogens
ER Tooltip Estrogen receptor agonists
Steroidal: Alfatradiol Certain androgens /anabolic steroids (e.g., testosterone , testosterone esters , methyltestosterone , metandienone , nandrolone esters ) (via estrogenic metabolites)
Certain progestins (e.g., norethisterone , noretynodrel , etynodiol diacetate , tibolone )
Clomestrone Cloxestradiol acetate Conjugated estriol Conjugated estrogens Epiestriol Epimestrol Esterified estrogens Estetrol † Estradiol Estradiol esters (e.g., estradiol acetate , estradiol benzoate , estradiol cypionate , estradiol enanthate , estradiol undecylate , estradiol valerate , polyestradiol phosphate , estradiol ester mixtures (Climacteron ))Estramustine phosphate Estriol Estriol esters (e.g., estriol succinate , polyestriol phosphate )Estrogenic substances Estrone Estrone esters
Ethinylestradiol #
Hydroxyestrone diacetate Mestranol Methylestradiol Moxestrol Nilestriol Prasterone (dehydroepiandrosterone; DHEA)
Promestriene Quinestradol Quinestrol Progonadotropins
Antiestrogens
ER Tooltip Estrogen receptor antagonistsSERMs Tooltip selective estrogen receptor modulators /SERDs Tooltip selective estrogen receptor downregulators )Aromatase inhibitors Antigonadotropins
Androgens /anabolic steroids (e.g., testosterone , testosterone esters , nandrolone esters , oxandrolone , fluoxymesterone )D2 receptor antagonists (prolactin releasers) (e.g., domperidone , metoclopramide , risperidone , haloperidol , chlorpromazine , sulpiride )GnRH agonistsleuprorelin , goserelin )GnRH antagonistscetrorelix , elagolix )Progestogens (e.g., chlormadinone acetate , cyproterone acetate , gestonorone caproate , hydroxyprogesterone caproate , medroxyprogesterone acetate , megestrol acetate ) Others
ER Tooltip Estrogen receptor
Agonists
Steroidal: 2-Hydroxyestradiol 2-Hydroxyestrone 3-Methyl-19-methyleneandrosta-3,5-dien-17β-ol 3α-Androstanediol 3α,5α-Dihydrolevonorgestrel 3β,5α-Dihydrolevonorgestrel 3α-Hydroxytibolone 3β-Hydroxytibolone 3β-Androstanediol 4-Androstenediol 4-Androstenedione 4-Fluoroestradiol 4-Hydroxyestradiol 4-Hydroxyestrone 4-Methoxyestradiol 4-Methoxyestrone 5-Androstenediol 7-Oxo-DHEA 7α-Hydroxy-DHEA 7α-Methylestradiol 7β-Hydroxyepiandrosterone 8,9-Dehydroestradiol 8,9-Dehydroestrone 8β-VE2 10β,17β-Dihydroxyestra-1,4-dien-3-one (DHED) 11β-Chloromethylestradiol 11β-Methoxyestradiol 15α-Hydroxyestradiol 16-Ketoestradiol 16-Ketoestrone 16α-Fluoroestradiol 16α-Hydroxy-DHEA 16α-Hydroxyestrone 16α-Iodoestradiol 16α-LE2 16β-Hydroxyestrone 16β,17α-Epiestriol (16β-hydroxy-17α-estradiol) 17α-Estradiol (alfatradiol )17α-Dihydroequilenin 17α-Dihydroequilin 17α-Epiestriol (16α-hydroxy-17α-estradiol) 17α-Ethynyl-3α-androstanediol 17α-Ethynyl-3β-androstanediol 17β-Dihydroequilenin 17β-Dihydroequilin 17β-Methyl-17α-dihydroequilenin Abiraterone Abiraterone acetate Alestramustine Almestrone Anabolic steroids (e.g., testosterone and esters , methyltestosterone , metandienone (methandrostenolone) , nandrolone and esters , many others; via estrogenic metabolites)Atrimustine Bolandiol Bolandiol dipropionate Butolame Clomestrone Cloxestradiol
Conjugated estriol Conjugated estrogens Cyclodiol Cyclotriol DHEA DHEA-S ent -EstradiolEpiestriol (16β-epiestriol, 16β-hydroxy-17β-estradiol) Epimestrol Equilenin Equilin ERA-63 (ORG-37663) Esterified estrogens Estetrol Estradiol
Estramustine Estramustine phosphate Estrapronicate Estrazinol Estriol
Estrofurate Estrogenic substances Estromustine Estrone
Etamestrol (eptamestrol) Ethinylandrostenediol
Ethinylestradiol
Ethinylestriol Ethylestradiol Etynodiol Etynodiol diacetate Hexolame Hippulin Hydroxyestrone diacetate Lynestrenol Lynestrenol phenylpropionate Mestranol Methylestradiol Moxestrol Mytatrienediol Nilestriol Norethisterone Noretynodrel Orestrate Pentolame Prodiame Prolame Promestriene RU-16117 Quinestradol Quinestrol Tibolone Xenoestrogens: Anise -related (e.g., anethole , anol , dianethole , dianol , photoanethole )Chalconoids (e.g., isoliquiritigenin , phloretin , phlorizin (phloridzin) , wedelolactone )Coumestans (e.g., coumestrol , psoralidin )Flavonoids (incl. 7,8-DHF , 8-prenylnaringenin , apigenin , baicalein , baicalin , biochanin A , calycosin , catechin , daidzein , daidzin , ECG , EGCG , epicatechin , equol , formononetin , glabrene , glabridin , genistein , genistin , glycitein , kaempferol , liquiritigenin , mirificin , myricetin , naringenin , penduletin , pinocembrin , prunetin , puerarin , quercetin , tectoridin , tectorigenin )Lavender oil Lignans (e.g., enterodiol , enterolactone , nyasol (cis -hinokiresinol) )Metalloestrogens (e.g., cadmium )Pesticides (e.g., alternariol , dieldrin , endosulfan , fenarimol , HPTE , methiocarb , methoxychlor , triclocarban , triclosan )Phytosteroids (e.g., digitoxin (digitalis ), diosgenin , guggulsterone )Phytosterols (e.g., β-sitosterol , campesterol , stigmasterol )Resorcylic acid lactones (e.g., zearalanone , α-zearalenol , β-zearalenol , zearalenone , zeranol (α-zearalanol) , taleranol (teranol, β-zearalanol) )Steroid -like (e.g., deoxymiroestrol , miroestrol )Stilbenoids (e.g., resveratrol , rhaponticin )Synthetic xenoestrogens (e.g., alkylphenols , bisphenols (e.g., BPA , BPF , BPS ), DDT , parabens , PBBs , PHBA , phthalates , PCBs )Others (e.g., agnuside , rotundifuran ) MixedSERMs Tooltip Selective estrogen receptor modulators ) Antagonists
Coregulator-binding modulators: ERX-11
GPER Tooltip G protein-coupled estrogen receptor
Agonists Antagonists Unknown