Beta-defensin 2
Beta-defensin 2 (BD-2) also known as skin-antimicrobial peptide 1 (SAP1) is a peptide that in humans is encoded by the DEFB4 (defensin, beta 4) gene.[3]
Human beta-defensin-2 (hBD-2) is a cysteine-rich cationic low molecular weight antimicrobial peptide discovered in lesional skin.
Structure
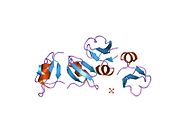
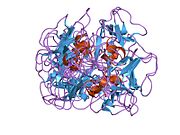
[edit]hBD-2 is a protein whose primary structure is made by 64 aminoacids. At concentrations ≤2.4 mM, hBD-2 is monomeric.[4] The structure is amphiphilic with a nonuniform surface distribution of positive charge and contains several key structural elements, including a triple-stranded, antiparallel beta sheet with strands 2 and 3 in a beta hairpin conformation. The determination of other structural elements depends on the technique used. When X-ray crystallography is used an alpha helix can be observed at the N-terminal end of the protein (PDB codes: 1fd3,1fd4, and 6cs9). When using NMR this alpha-helix does not appear (PDB code: 1e4q), however this structure was determined using a truncated version of hBD-2 which was missing the initial 4 amino acids, and may be the reason for the discrepancy.
Function
[edit]Defensins form a family of microbicidal and cytotoxic peptides made by neutrophils. Members of the defensin family are highly similar in protein sequence. Beta-defensin 2 is an antibiotic peptide which is locally regulated by inflammation.[5]
Human beta-defensin 2 is produced by a number of epithelial cells and exhibits potent antimicrobial activity against Gram-negative bacteria and Candida, but not Gram-positive S. aureus. It has been speculated that beta-defensin 2 may contribute to the infrequency of Gram-negative infections on skin and lung tissue.[6]
hBD-2 represents the first human defensin that is produced following stimulation of epithelial cells by contact with microorganisms such as P. aeruginosa or cytokines such as TNF-alpha and IL-1 beta. The HBD-2 gene and protein are locally expressed in keratinocytes associated with inflammatory skin lesions. It is intriguing to speculate that HBD-2 is a dynamic component of the local epithelial defense system of the skin and respiratory tract having a role to protect surfaces from infection, and providing a possible reason why skin and lung infections with Gram-negative bacteria are rather rare.[6]
Although this protein doesn’t have any antibacterial activity against Gram-positive bacteria, there is a study showing that there is a synergy between hBD-2 and other proteins.[7] One example of this synergistic effect is with epiP, a protein segregated by some strains of S. epidermidis. hBD2, holding hands with epiP, is capable of killing S. aureus, a Gram-positive bacteria responsible of human diseases.
References
[edit]- ^ a b c ENSG00000275444, ENSG00000285433 GRCh38: Ensembl release 89: ENSG00000177257, ENSG00000275444, ENSG00000285433 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Harder J, Bartels J, Christophers E, Schröder JM (June 1997). "A peptide antibiotic from human skin". Nature. 387 (6636): 861. doi:10.1038/43088. PMID 9202117. S2CID 4354862.
- ^ Sawai MV, Jia HP, Liu L, Aseyev V, Wiencek JM, McCray PB, Ganz T, Kearney WR, Tack BF (April 2001). "The NMR structure of human beta-defensin-2 reveals a novel alpha-helical segment". Biochemistry. 40 (13): 3810–3816. doi:10.1021/bi002519d. PMID 11300761.
- ^ "Entrez Gene: DEFB4 defensin, beta 4".
- ^ a b Schröder JM, Harder J (June 1999). "Human beta-defensin-2". The International Journal of Biochemistry & Cell Biology. 31 (6): 645–651. doi:10.1016/S1357-2725(99)00013-8. PMID 10404637.
- ^ Iwase T, Uehara Y, Shinji H, Tajima A, Seo H, Takada K, Agata T, Mizunoe Y (May 2010). "Staphylococcus epidermidis Esp inhibits Staphylococcus aureus biofilm formation and nasal colonization". Nature. 465 (7296): 346–349. Bibcode:2010Natur.465..346I. doi:10.1038/nature09074. PMID 20485435. S2CID 4392908.
External links
[edit]- Human DEFB4A genome location and DEFB4A gene details page in the UCSC Genome Browser.
Further reading
[edit]- Weinberg A, Krisanaprakornkit S, Dale BA (1999). "Epithelial antimicrobial peptides: review and significance for oral applications". Critical Reviews in Oral Biology and Medicine. 9 (4): 399–414. doi:10.1177/10454411980090040201. PMID 9825219.
- Schröder JM, Harder J (June 1999). "Human beta-defensin-2". The International Journal of Biochemistry & Cell Biology. 31 (6): 645–651. doi:10.1016/S1357-2725(99)00013-8. PMID 10404637.
- Bals R, Wang X, Wu Z, Freeman T, Bafna V, Zasloff M, Wilson JM (September 1998). "Human beta-defensin 2 is a salt-sensitive peptide antibiotic expressed in human lung". The Journal of Clinical Investigation. 102 (5): 874–880. doi:10.1172/JCI2410. PMC 508952. PMID 9727055.
- Liu L, Wang L, Jia HP, Zhao C, Heng HH, Schutte BC, McCray PB, Ganz T (November 1998). "Structure and mapping of the human beta-defensin HBD-2 gene and its expression at sites of inflammation". Gene. 222 (2): 237–244. doi:10.1016/S0378-1119(98)00480-6. PMID 9831658.
- Mathews M, Jia HP, Guthmiller JM, Losh G, Graham S, Johnson GK, Tack BF, McCray PB (June 1999). "Production of beta-defensin antimicrobial peptides by the oral mucosa and salivary glands". Infection and Immunity. 67 (6): 2740–2745. doi:10.1128/IAI.67.6.2740-2745.1999. PMC 96577. PMID 10338476.
- Jia HP, Mills JN, Barahmand-Pour F, Nishimura D, Mallampali RK, Wang G, Wiles K, Tack BF, Bevins CL, McCray PB (September 1999). "Molecular cloning and characterization of rat genes encoding homologues of human beta-defensins". Infection and Immunity. 67 (9): 4827–4833. doi:10.1128/IAI.67.9.4827-4833.1999. PMC 96815. PMID 10456937.
- Yang D, Chertov O, Bykovskaia SN, Chen Q, Buffo MJ, Shogan J, Anderson M, Schröder JM, Wang JM, Howard OM, Oppenheim JJ (October 1999). "Beta-defensins: linking innate and adaptive immunity through dendritic and T cell CCR6". Science. 286 (5439): 525–528. doi:10.1126/science.286.5439.525. PMID 10521347.
- Diamond G, Kaiser V, Rhodes J, Russell JP, Bevins CL (January 2000). "Transcriptional regulation of beta-defensin gene expression in tracheal epithelial cells". Infection and Immunity. 68 (1): 113–119. doi:10.1128/iai.68.1.113-119.2000. PMC 97109. PMID 10603376.
- Harder J, Meyer-Hoffert U, Teran LM, Schwichtenberg L, Bartels J, Maune S, Schröder JM (June 2000). "Mucoid Pseudomonas aeruginosa, TNF-alpha, and IL-1beta, but not IL-6, induce human beta-defensin-2 in respiratory epithelia". American Journal of Respiratory Cell and Molecular Biology. 22 (6): 714–721. doi:10.1165/ajrcmb.22.6.4023. PMID 10837369.
- Hoover DM, Rajashankar KR, Blumenthal R, Puri A, Oppenheim JJ, Chertov O, Lubkowski J (October 2000). "The structure of human beta-defensin-2 shows evidence of higher order oligomerization". The Journal of Biological Chemistry. 275 (42): 32911–32918. doi:10.1074/jbc.M006098200. PMID 10906336.
- Meyer JE, Harder J, Görögh T, Schröder JM, Maune S (July 2000). "[hBD-2 gene expression in nasal mucosa]". Laryngo- Rhino- Otologie. 79 (7): 400–403. doi:10.1055/s-2000-4626. PMID 11005092. S2CID 71240506.
- Ali RS, Falconer A, Ikram M, Bissett CE, Cerio R, Quinn AG (July 2001). "Expression of the peptide antibiotics human beta defensin-1 and human beta defensin-2 in normal human skin". The Journal of Investigative Dermatology. 117 (1): 106–111. doi:10.1046/j.0022-202x.2001.01401.x. PMID 11442756.
- Carothers DG, Graham SM, Jia HP, Ackermann MR, Tack BF, McCray PB (2001). "Production of beta-defensin antimicrobial peptides by maxillary sinus mucosa". American Journal of Rhinology. 15 (3): 175–179. doi:10.2500/105065801779954238. PMID 11453504. S2CID 31583849.
- Biragyn A, Surenhu M, Yang D, Ruffini PA, Haines BA, Klyushnenkova E, Oppenheim JJ, Kwak LW (December 2001). "Mediators of innate immunity that target immature, but not mature, dendritic cells induce antitumor immunity when genetically fused with nonimmunogenic tumor antigens". Journal of Immunology. 167 (11): 6644–6653. doi:10.4049/jimmunol.167.11.6644. PMID 11714836.
- Bauer F, Schweimer K, Klüver E, Conejo-Garcia JR, Forssmann WG, Rösch P, Adermann K, Sticht H (December 2001). "Structure determination of human and murine beta-defensins reveals structural conservation in the absence of significant sequence similarity". Protein Science. 10 (12): 2470–2479. doi:10.1110/ps.24401. PMC 2374044. PMID 11714914.
- Takahashi A, Wada A, Ogushi K, Maeda K, Kawahara T, Mawatari K, Kurazono H, Moss J, Hirayama T, Nakaya Y (November 2001). "Production of beta-defensin-2 by human colonic epithelial cells induced by Salmonella enteritidis flagella filament structural protein". FEBS Letters. 508 (3): 484–488. doi:10.1016/S0014-5793(01)03088-5. PMID 11728477. S2CID 24925463.
- Schibli DJ, Hunter HN, Aseyev V, Starner TD, Wiencek JM, McCray PB, Tack BF, Vogel HJ (March 2002). "The solution structures of the human beta-defensins lead to a better understanding of the potent bactericidal activity of HBD3 against Staphylococcus aureus". The Journal of Biological Chemistry. 277 (10): 8279–8289. doi:10.1074/jbc.M108830200. PMID 11741980.