Atmospheric methane removal
Atmospheric methane removal is a category of potential approaches being researched to accelerate the breakdown of methane that is in the atmosphere, for the purpose of mitigating some of the impacts of climate change.[1]
Atmospheric methane has increased since pre-industrial times from 0.7 ppm to 1.9 ppm.[2] From 2010 to 2019, methane emissions caused 0.5 °C (about 30%) of observed global warming.[3][4] Global methane emissions approached a record 600 Tg CH4 per year in 2017.[1]
Natural atmospheric methane sinks
[edit]
Methane has a limited atmospheric lifetime, about 10 years, due to substantial methane sinks. The primary methane sink is atmospheric oxidation, from hydroxyl radicals (~90% of the total sink) and chlorine radicals (0-5% of the total sink). The rest is consumed by methanotrophs and other methane-oxidizing bacteria and archaea in soils (~5%).[5]
Potential approaches
[edit]Different methods to remove methane from the atmosphere include thermal-catalytic oxidation, photocatalytic oxidation, biological methanotrophic methane removal, concentration with zeolites or other porous solids, and separation by membranes.[6]
Potential methods can be categorized by the underlying catalytic process, or the potential deployment form.
Enhanced atmospheric methane oxidation
[edit]Enhanced Atmospheric Methane Oxidation is the concept of enhancing the overall oxidative methane sink in the atmosphere, through generating additional hydroxyl or chlorine atmospheric radicals.
Iron salt aerosols
[edit]Iron salt aerosols are one proposed method of enhanced atmospheric methane oxidation which involves lofting iron-based particles into the atmosphere (e.g. from planes[7] or ships) to enhance atmospheric chlorine radicals, a natural methane sink.[8] Winds over the Sahara raise dust into the troposphere and disperse it over the Atlantic.[9] A 2023 study suggests that this has contributed to natural atmospheric methane oxidation.[10][11]
Iron salt aerosols are being studied for the potential of iron(III) chloride (FeCl3) to catalyze chlorine radical production.[12] Chlorine atoms are produced by photolysis from the FeCl3 stemming from iron-containing airborne dust aerosol particles in the oceanic boundary layer.[13]
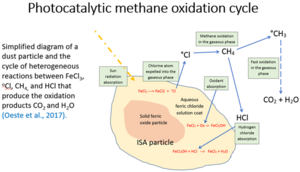
- FeCl3 + hv → FeCl2 + oCl
The chlorine atoms initiate methane oxidation:
- CH4 + oCl → HCl + oCH3
The resulting methyl radical is unstable and oxidises naturally to CO2 and water:
- 3.5O2 + 2oCH3 → 2CO2 + 3H2O
Side effects of ferric chloride
[edit]Fine particles dispersed in the atmosphere can serve as cloud condensation nuclei and thereby cause marine cloud brightening[14]
Eventually all FeCl3 particles are washed out of the air and fall on land or water, where they dissolve into iron compounds and salt.[12]
Iron salt aerosols may also therefore contribute to iron fertilization.
Terrestrial methanotroph enhancement
[edit]Soil bacteria and archaea account for approximately 5% of the natural methane sink. Early research is going into how the activity of these bacteria may be able to be enhanced, either through the use of soil amendments, or introduction of selected or engineered methane-oxidizing bacteria.[15]
Catalytic engineered systems
[edit]Catalytic engineered systems are designed to pass air from the atmosphere, either passively or actively, through catalytic systems which leverage energy from the sun, an artificial light, or heat to oxidize methane. These catalysts include thermocatalysts, photocatalysts, and radicals produced artificially through photolysis (using light to break apart a molecule).[15]
References
[edit]- ^ a b Jackson, Robert (2021). "Atmospheric methane removal: a research agenda". Philosophical Transactions A. 379 (20200454). Bibcode:2021RSPTA.37900454J. doi:10.1098/rsta.2020.0454. PMC 8473948. PMID 34565221.
- ^ "Rising methane could be a sign that Earth's climate is part-way through a 'termination-level transition'". 14 Aug 2023.
- ^ "Figure AR6 WG1". ipcc.ch. Retrieved 2023-10-05.
- ^ "Methane and climate change".
- ^ Saunois, Marielle; Stavert, Ann R.; Poulter, Ben; Bousquet, Philippe; Canadell, Josep G.; Jackson, Robert B.; Raymond, Peter A.; Dlugokencky, Edward J.; Houweling, Sander; Patra, Prabir K.; Ciais, Philippe; Arora, Vivek K.; Bastviken, David; Bergamaschi, Peter; Blake, Donald R. (2020-07-15). "The Global Methane Budget 2000–2017". Earth System Science Data. 12 (3): 1561–1623. Bibcode:2020ESSD...12.1561S. doi:10.5194/essd-12-1561-2020. ISSN 1866-3508.
- ^ Nisbet-Jones, Peter B. R.; Fernandez, Julianne M.; Fisher, Rebecca E.; France, James L.; Lowry, David; Waltham, David A.; Woolley Maisch, Ceres A.; Nisbet, Euan G. (24 January 2022). "Is the destruction or removal of atmospheric methane a worthwhile option?". Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences. 380 (2215). Bibcode:2022RSPTA.38010108N. doi:10.1098/rsta.2021.0108. PMC 8646139. PMID 34865528.
- ^ Atmospheric Methane Removal AG (March 8, 2024). "Dispersion by planes". amr.earth.
- ^ "Iron Salt Aerosols — Atmospheric Methane Removal". sparkclimate.org. Retrieved 2024-04-09.
- ^ Radford, Tim (July 16, 2014). "Desert Dust Feeds Deep Ocean Life". The Daily Climate.
- ^ "Spark Climate Solutions". sparkclimate.org. Retrieved 2023-10-05.
- ^ van Herpen, Maarten; et al. (2023). "Photocatalytic chlorine atom production on mineral dust–sea spray aerosols over the North Atlantic". PNAS. 120 (31): e2303974120. Bibcode:2023PNAS..12003974V. doi:10.1073/pnas.2303974120. PMC 10400977. PMID 37487065.
- ^ a b Franz D. Oeste; Renaud de Richter; Thingzhen Ming (2017). "Climate engineering by mimicking natural dust climate control: the iron salt aerosol method". Earth System Dynamics. 8 (1): 1–54. Bibcode:2017ESD.....8....1O. doi:10.5194/esd-8-1-2017.
- ^ Oum KW, Lakin MJ, DeHaan DO, Brauers T, Finlayson-Pitts BJ (2 Jan 1998). "Formation of molecular chlorine from the photolysis of ozone and aqueous sea-salt particles". Science. 279 (5347): 74–77. Bibcode:1998Sci...279...74O. doi:10.1126/science.279.5347.74. PMID 9417027.
- ^ Mace, Gerald G. (1 Feb 2023). "Natural marine cloud brightening in the Southern Ocean". EGU Atmospheric Chemistry and Physics. 23 (1677–1685): 74–77. doi:10.5194/acp-23-1677-2023.
- ^ a b Jackson, Robert B.; Abernethy, Sam; Canadell, Josep G.; Cargnello, Matteo; Davis, Steven J.; Féron, Sarah; Fuss, Sabine; Heyer, Alexander J.; Hong, Chaopeng; Jones, Chris D.; Damon Matthews, H.; O'Connor, Fiona M.; Pisciotta, Maxwell; Rhoda, Hannah M.; de Richter, Renaud (2021-11-15). "Atmospheric methane removal: a research agenda". Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences. 379 (2210): 20200454. Bibcode:2021RSPTA.37900454J. doi:10.1098/rsta.2020.0454. ISSN 1364-503X. PMC 8473948. PMID 34565221.
