Bosentan
 | |
| Clinical data | |
|---|---|
| Trade names | Tracleer, Stayveer, Safebo |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a605001 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 50% |
| Protein binding | >98% |
| Metabolism | Liver |
| Elimination half-life | 5 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.171.206 |
| Chemical and physical data | |
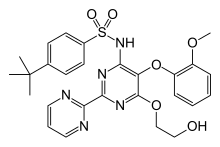
| Formula | C27H29N5O6S |
| Molar mass | 551.62 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Bosentan, sold under the brand name Tracleer among others, is a dual endothelin receptor antagonist medication used in the treatment of pulmonary artery hypertension (PAH).[4][5]
Bosentan is available as film-coated tablets (62.5 mg or 125 mg) or as dispersable tablets for oral suspension (32 mg).[4]
Medical uses
[edit]Bosentan is used to treat people with moderate pulmonary arterial hypertension and to reduce the number of digital ulcers — open wounds on especially on fingertips and less commonly the knuckles — in people with systemic scleroderma.[4][3][6]
Contraindications
[edit]Bosentan is contraindicated in people taking glyburide due to an increased risk of increased liver enzymes and liver damage when these two agents are taken together.[4]
Use of bosentan with cyclosporine is contraindicated because cyclosporine A has been shown to markedly increase serum concentration of bosentan.[4]
Adverse effects
[edit]Bosentan causes harm to fetuses (teratogenic) and it may render hormonal contraceptives ineffective.[4][3]
In the US it is only available from doctors who follow an FDA-mandated risk evaluation and mitigation strategy (REMS) with respect to risks to fetuses and its risks of causing liver damage.[7]
In addition to the risk of causing birth defects and of causing liver damage, bosentan has a high risk of causing edema, pulmonary veno-occlusive disease, decreasing sperm counts, and decreases in hemoglobin and hematocrit.[4][3]
Very common adverse effects (occurring in more than 10% of people) include headache, elevated transaminases, and edema. Common adverse effects (between 1% and 10% of people) include anemia, reduced hemoglobin, hypersensitivity reactions, skin inflammation, itchiness, rashes, red skin, flushing, fainting, heart palpitations, low blood pressure, nasal congestion, gastro-esophageal reflux disease, and diarrhea.[4][3]
Drug interactions
[edit]Bosentan may render hormonal contraceptives ineffective.[4][3]
Mechanism of action
[edit]Bosentan is a competitive antagonist of endothelin-1 at the endothelin-A (ET-A) and endothelin-B (ET-B) receptors. Under normal conditions, endothelin-1 binding of ET-A receptors causes constriction of the pulmonary blood vessels.[8] Conversely, binding of endothelin-1 to ET-B receptors has been associated with both vasodilation and vasoconstriction of vascular smooth muscle, depending on the ET-B subtype (ET-B1 or ET-B2) and tissue.[9] Bosentan blocks both ET-A and ET-B receptors, but is thought to exert a greater effect on ET-A receptors, causing a total decrease in pulmonary vascular resistance.[4]
Pharmacokinetics
[edit]After oral administration, maximum plasma concentrations of bosentan are attained within 3–5 hours and the terminal elimination half-life (t1/2) is about 5 hours in healthy adult subjects. The exposure to bosentan after intervenous and oral administration is about 2-fold greater in adult patients with pulmonary arterial hypertension than in healthy adult subjects.[10]
Absolute bioavailability of bosentan is about 50% in healthy subjects.[11] Peak plasma concentration of bosentan with the dispersable tablets for oral suspension is 14% less on average compared to peak concentration of the oral tablets.[4]
Bosentan is a substrate of CYP3A4 and CYP2C9. CYP2C19 may also play a role in its metabolism.[4] It is also a substrate of the hepatic uptake transporter organic anion-transporting polypeptides (OATPs) OATP1B1, OATP1B3, and OATP2B1.[12][13]
Elimination of bosentan is mostly hepatic, with minimal contribution from renal and fecal excretion.[14]
Use of bosentan with cyclosporine is contraindicated because cyclosporine A has been shown to markedly increase serum concentration of bosentan.[4]
History
[edit]Bosentan was studied in heart failure in a trial called REACH-1 that was terminated early in 1997, due to toxicity at the dose that was being studied. [15]
It was approved for pulmonary artery hypertension in the US in November 2001,[4][16] and in the European Union in May 2002.[3][5]
Society and culture
[edit]Economics
[edit]By 2013, worldwide sales of bosentan were $1.57 billion. The patents on bosentan started expiring in 2015.[17]
References
[edit]- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ "Prescription medicines: registration of new generic medicines and biosimilar medicines, 2017". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 30 March 2024.
- ^ a b c d e f g "Tracleer (bosentan) 62.5 mg and 125mg film-coated tablets". UK Electronic Medicines Compendium. May 2017. Archived from the original on 27 July 2020. Retrieved 6 August 2017.
- ^ a b c d e f g h i j k l m n o "Tracleer- bosentan tablet, film coated Tracleer- bosentan tablet, soluble". DailyMed. 15 June 2020. Retrieved 15 October 2020.
- ^ a b c "Tracleer EPAR". European Medicines Agency (EMA). 17 September 2018. Retrieved 16 October 2020.
- ^ Abraham S, Steen V (2015). "Optimal management of digital ulcers in systemic sclerosis". Therapeutics and Clinical Risk Management. 11: 939–47. doi:10.2147/TCRM.S82561. PMC 4474386. PMID 26109864.
- ^ "Approved Risk Evaluation and Mitigation Strategies (REMS)". U.S. Food and Drug Administration (FDA). Retrieved 6 August 2017.
- ^ Givertz MM, Colucci WS, LeJemtel TH, Gottlieb SS, Hare JM, Slawsky MT, et al. (June 2000). "Acute endothelin A receptor blockade causes selective pulmonary vasodilation in patients with chronic heart failure". Circulation. 101 (25): 2922–7. doi:10.1161/01.CIR.101.25.2922. PMID 10869264.
- ^ Hynynen MM, Khalil RA (January 2006). "The vascular endothelin system in hypertension--recent patents and discoveries". Recent Patents on Cardiovascular Drug Discovery. 1 (1): 95–108. doi:10.2174/157489006775244263. PMC 1351106. PMID 17200683.
- ^ "patient information leaflets"
- ^ Weber C, Schmitt R, Birnboeck H, Hopfgartner G, van Marle SP, Peeters PA, et al. (August 1996). "Pharmacokinetics and pharmacodynamics of the endothelin-receptor antagonist bosentan in healthy human subjects". Clinical Pharmacology and Therapeutics. 60 (2): 124–37. doi:10.1016/S0009-9236(96)90127-7. PMID 8823230. S2CID 3039181.
- ^ Jones HM, Barton HA, Lai Y, Bi YA, Kimoto E, Kempshall S, et al. (May 2012). "Mechanistic pharmacokinetic modeling for the prediction of transporter-mediated disposition in humans from sandwich culture human hepatocyte data". Drug Metabolism and Disposition. 40 (5): 1007–17. doi:10.1124/dmd.111.042994. PMID 22344703. S2CID 15463540.
- ^ Treiber A, Schneiter R, Häusler S, Stieger B (August 2007). "Bosentan is a substrate of human OATP1B1 and OATP1B3: inhibition of hepatic uptake as the common mechanism of its interactions with cyclosporin A, rifampicin, and sildenafil". Drug Metabolism and Disposition. 35 (8): 1400–7. doi:10.1124/dmd.106.013615. PMID 17496208. S2CID 2625368.
- ^ Weber C, Gasser R, Hopfgartner G (July 1999). "Absorption, excretion, and metabolism of the endothelin receptor antagonist bosentan in healthy male subjects". Drug Metabolism and Disposition. 27 (7): 810–5. PMID 10383925.
- ^ Packer M, McMurray J, Massie BM, Caspi A, Charlon V, Cohen-Solal A, et al. (February 2005). "Clinical effects of endothelin receptor antagonism with bosentan in patients with severe chronic heart failure: results of a pilot study". Journal of Cardiac Failure. 11 (1): 12–20. doi:10.1016/j.cardfail.2004.05.006. PMID 15704058.
- ^ "Drug Approval Package: Tracleer (Bosentan) NDA #21-290". U.S. Food and Drug Administration (FDA). 20 November 2001. Retrieved 16 October 2020.
- ^ Helfand C (2015). "The top 10 patent losses of 2015: Tracleer". FiercePharma.
