Nedocromil
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Alocril |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601243 |
| Routes of administration | Inhalation and eye drops |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 89% |
| Metabolism | not metabolized |
| Elimination half-life | ~3.3 hours |
| Excretion | excreted unchanged |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.233.208 |
| Chemical and physical data | |
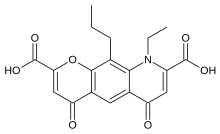
| Formula | C19H17NO7 |
| Molar mass | 371.345 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Nedocromil sodium is a medication considered as mast cell stabilizer which acts to prevent wheezing, shortness of breath, and other breathing problems caused by asthma. It is administered by an inhaler under the brand name Tilade, and as an eye drop under the brand name Alocril.[1][2] The effects of nedocromil versus asthma are gradual rather than fast-acting and it is not indicated for acute respiratory distress compared to fast acting bronchodilators like albuterol or other well-known inhaler medications. Liquid preparations of nedocromil are available in the UK under the name Rapitil for use for allergic eye reactions.[3] Nedocromil sodium has been shown to be effective in alleviating symptoms of allergic conjunctivitis.[4]
Nedocromil is classified as a benzopyrone. Nedocromil acts as a mast cell stabilizer, inhibits the degranulation of mast cells, prevents release of histamine and tryptase, so preventing the synthesis of prostaglandins and leukotrienes. US Production of inhaled nedocromil ceased in April 2008 because it used CFCs as propellant.[5]
See also
[edit]References
[edit]![]() Media related to Nedocromil at Wikimedia Commons
Media related to Nedocromil at Wikimedia Commons
- ^ "ALOCRIL Product Information". Allergan. Archived from the original on 8 May 2013. Retrieved 17 May 2013.
- ^ Allen H. Dajani S (ed.). "ALOCRIL (nedocromil sodium) solution/ drops". DailyMed. U.S. National Institutes of Health. Retrieved 17 May 2013.
- ^ "Nedocromil eye drops". Patient.info. 2007-02-02. Retrieved 2009-08-04.
- ^ Castillo M, Scott NW, Mustafa MZ, Mustafa MS, Azuara-Blanco A (June 2015). "Topical antihistamines and mast cell stabilisers for treating seasonal and perennial allergic conjunctivitis" (PDF). The Cochrane Database of Systematic Reviews. 2015 (6): CD009566. doi:10.1002/14651858.CD009566.pub2. hdl:2164/6048. PMC 10616535. PMID 26028608.
- ^ "Questions and Answers:Phase-Out of CFC Metered-Dose Inhalers Containing flunisolide, triamcinolone, metaproterenol, pirbuterol, albuterol and ipratropium in combination, cromolyn, and nedocromil". U.S. Food and Drug Administration. 13 April 2010. Archived from the original on 2 February 2012.
