Sakurai reaction
| Sakurai reaction | |
|---|---|
| Named after | Hideki Sakurai Akira Hosomi |
| Reaction type | Addition reaction |
| Identifiers | |
| Organic Chemistry Portal | hosomi-sakurai-reaction |
| RSC ontology ID | RXNO:0000443 |
The Sakurai reaction (also known as the Hosomi–Sakurai reaction) is the chemical reaction of carbon electrophiles (such as a ketone shown here) with allyltrimethylsilane catalyzed by strong Lewis acids.[1][2][3] The reaction achieves results similar to the addition of an allyl Grignard reagent to the carbonyl.
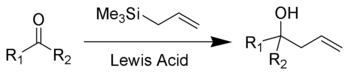
Strong Lewis acids such as titanium tetrachloride, boron trifluoride, tin tetrachloride, and AlCl(Et)2 are all effective in promoting the Hosomi reaction. The reaction involves electrophilic allyl shift via a beta-silyl carbocationic intermediate, the beta-silicon effect.[4]
Mechanism
[edit]

Allylation of a carbonyl ketone (compound containing a ketone group and two different functional groups) has been shown. In the given reaction, the electrophilic compound (carbon with a ketone group) is treated with titanium tetrachloride, a strong Lewis acid and allyltrimethylsilane. According to the general principle, the Lewis acid first activates the electrophilic carbon in presence of allyltrimethylsilane which then undergoes nucleophilic attack from electrons on the allylic silane.[5] The silicon plays the key role in stabilizing the carbocation of carbon at the β-position. Hosomi-Sakurai reaction is also applicable for other functional groups such as enones, where conjugate addition is usually seen. In figure 2, the Hosomi- Sakurai reaction has been shown using a cinnamoyl ketone. This reaction follows the same mechanism as the previous reaction shown here.
As displayed in the scheme, the Hosomi–Sakurai reaction is proposed to give a secondary carbocation intermediate. Secondary carbocations are high in energy, however it is stabilized by the silicon substituent ("β-silicon effect", a form of silicon-hyperconjugation).
Literature of historic interest
[edit]- Sakurai, Hideki; Hosomi, Akira; Kumada, Makoto (1969). "Addition of trichloromethyl radicals to alkenylsilanes". The Journal of Organic Chemistry. 36 (4): 1764–1768. doi:10.1021/jo01258a052.
- Hosomi, Akíra; Sakurai, Hideki (1976). "Syntheses of γ,δ-unsaturated alcohols from allylsilanes and carbonyl compounds in the presence of titanium tetrachloride". Tetrahedron Letters. 17 (16): 1295–1298. doi:10.1016/S0040-4039(00)78044-0. ISSN 0040-4039.
- (Hosomi, Akira; Endo, Masahiko; Sakurai, Hideki (5 September 1976). "Allylsilanes as synthetic intermediates. ii. syntheses of homoallyl ethers from allylsilanes and acetals promoted by titanium tetrachloride". Chemistry Letters. 5 (9): 941–942. doi:10.1246/cl.1976.941. ISSN 0366-7022.
- Hosomi, Akira; Sakurai, Hideki (1 March 1977). "Chemistry of organosilicon compounds. 99. Conjugate addition of allylsilanes to .alpha.,.beta.-enones. A New method of stereoselective introduction of the angular allyl group in fused cyclic .alpha.,.beta.-enones". Journal of the American Chemical Society. 99 (5): 1673–1675. doi:10.1021/ja00447a080. ISSN 0002-7863.
References
[edit]- ^ Hosomi, Akira (1 May 1988). "Characteristics in the reactions of allylsilanes and their applications to versatile synthetic equivalents". Accounts of Chemical Research. 21 (5): 200–206. doi:10.1021/ar00149a004. ISSN 0001-4842.
- ^ Fleming, Ian; Dunoguès, Jacques; Smithers, Roger (2004), "The Electrophilic Substitution of Allylsilanes and Vinylsilanes", Organic Reactions, American Chemical Society, pp. 57–575, doi:10.1002/0471264180.or037.02, ISBN 9780471264187
- ^ Fleming, Ian (1 January 1991). "Allylsilanes, Allylstannanes and Related Systems". In Trost, Barry M.; Fleming, Ian (eds.). 2.2 – Allylsilanes, Allylstannanes and Related Systems. Pergamon. pp. 563–593. doi:10.1016/b978-0-08-052349-1.00041-x. ISBN 9780080523491.
{{cite book}}:|work=ignored (help) - ^ "Hosomi-Sakurai Reaction". organic-chemistry.org. Retrieved 1 September 2019.
- ^ Yamasaki, Shingo; Fujii, Kunihiko; Wada, Reiko; Kanai, Motomu; Shibasaki, Masakatsu (1 June 2002). "A General Catalytic Allylation Using Allyltrimethoxysilane". Journal of the American Chemical Society. 124 (23): 6536–6537. doi:10.1021/ja0262582. ISSN 0002-7863. PMID 12047165.
