Gamma-glutamyl carboxylase
| GGCX | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | GGCX, VKCFD1, gamma-glutamyl carboxylase, Gamma-glutamyl carboxylase; GGCX | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 137167; MGI: 1927655; HomoloGene: 639; GeneCards: GGCX; OMA:GGCX - orthologs | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Gamma-glutamyl carboxylase is an enzyme that in humans is encoded by the GGCX gene, located on chromosome 2 at 2p12.[4]
Function
[edit]Gamma-glutamyl carboxylase is an enzyme that catalyzes the posttranslational modification of vitamin K-dependent proteins. Many of these vitamin K-dependent proteins are involved in coagulation so the function of the encoded enzyme is essential for hemostasis.[5] Most gla domain-containing proteins depend on this carboxylation reaction for posttranslational modification.[6] In humans, the gamma-glutamyl carboxylase enzyme is most highly expressed in the liver.
Catalytic reaction
[edit]Gamma-glutamyl carboxylase oxidizes vitamin K hydroquinone to Vitamin K-2,3-epoxide, while simultaneously adding CO2 to protein-bound glutamic acid (abbreviation = Glu) to form gamma-carboxyglutamic acid (also called gamma-carboxyglutamate, abbreviation = Gla). Presence of two carboxylate groups causes chelation of Ca2+, resulting in change in tertiary structure of protein and its activation. The carboxylation reaction will only proceed if the carboxylase enzyme is able to oxidize vitamin K hydroquinone to vitamin K epoxide at the same time; the carboxylation and epoxidation reactions are said to be coupled reactions.[7][8]
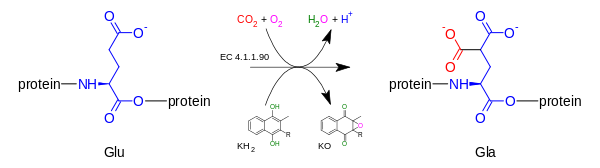
2) + CO
2 + oxygen → a [protein] 4-carboxy-L-glutamate (Gla) + vitamin K 2,3-epoxide (KO) + H+
+ H
2O
No experimental structure is known for GGCX, limiting understanding of its reaction mechanism. Based on the fact that the two reactions are coupled, a computational study is able to propose how the reactants interact with each other to form the products.[9] Lys228 has been shown to be the residue responsible for starting the reaction.[10] How the enzyme holds the reactants in place to have them interact with each other remains poorly shown. 491-507 and 395-401 are probably responsible for propeptide and glutamate binding respectively.[11]
Clinical significance
[edit]Mutations in this gene are associated with vitamin K-dependent coagulation defect and PXE-like disorder with multiple coagulation factor deficiency.[5][12]
See also
[edit]References
[edit]- ^ a b c GRCh38: Ensembl release 89: ENSG00000115486 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Wu SM, Cheung WF, Frazier D, Stafford DW (December 1991). "Cloning and expression of the cDNA for human gamma-glutamyl carboxylase". Science. 254 (5038): 1634–6. Bibcode:1991Sci...254.1634W. doi:10.1126/science.1749935. PMID 1749935.
- ^ a b "Entrez Gene: GGCX".
- ^ Brenner B, Tavori S, Zivelin A, Keller CB, Suttie JW, Tatarsky I, Seligsohn U (August 1990). "Hereditary deficiency of all vitamin K-dependent procoagulants and anticoagulants". Br. J. Haematol. 75 (4): 537–42. doi:10.1111/j.1365-2141.1990.tb07795.x. PMID 2145029. S2CID 24679257.
- ^ Suttie JW (1985). "Vitamin K-dependent carboxylase". Annu. Rev. Biochem. 54 (1): 459–77. doi:10.1146/annurev.bi.54.070185.002331. PMID 3896125.
- ^ Presnell SR, Stafford DW (2002). "The vitamin K-dependent carboxylase". Thromb. Haemost. 87 (6): 937–46. doi:10.1055/s-0037-1613115. PMID 12083499. S2CID 27634025.
- ^ Silva PJ, Ramos MJ (2007). "Reaction mechanism of the vitamin K-dependent glutamate carboxylase: a computational study". J Phys Chem B. 111 (44): 12883–7. doi:10.1021/jp0738208. PMID 17935315.
- ^ Rishavy MA, Hallgren KW, Yakubenko AV, Shtofman RL, Runge KW, Berkner KL (7 November 2006). "Brønsted analysis reveals Lys218 as the carboxylase active site base that deprotonates vitamin K hydroquinone to initiate vitamin K-dependent protein carboxylation". Biochemistry. 45 (44): 13239–48. doi:10.1021/bi0609523. PMID 17073445.
- ^ Parker CH, Morgan CR, Rand KD, Engen JR, Jorgenson JW, Stafford DW (11 March 2014). "A conformational investigation of propeptide binding to the integral membrane protein γ-glutamyl carboxylase using nanodisc hydrogen exchange mass spectrometry". Biochemistry. 53 (9): 1511–20. doi:10.1021/bi401536m. PMC 3970815. PMID 24512177.
- ^ Vanakker OM, Martin L, Gheduzzi D, Leroy BP, Loeys BL, Guerci VI, Matthys D, Terry SF, Coucke PJ, Pasquali-Ronchetti I, De Paepe A (March 2007). "Pseudoxanthoma elasticum-like phenotype with cutis laxa and multiple coagulation factor deficiency represents a separate genetic entity". J. Invest. Dermatol. 127 (3): 581–7. doi:10.1038/sj.jid.5700610. PMID 17110937.
Further reading
[edit]- Bandyopadhyay PK (2008). "Vitamin K-dependent gamma-glutamylcarboxylation: an ancient posttranslational modification". Vitam. Horm. Vitamins & Hormones. 78: 157–84. doi:10.1016/S0083-6729(07)00008-8. ISBN 9780123741134. PMID 18374194.
- Berkner KL (2008). "Vitamin K-dependent carboxylation". Vitam. Horm. Vitamins & Hormones. 78: 131–56. doi:10.1016/S0083-6729(07)00007-6. ISBN 9780123741134. PMID 18374193.
- Oldenburg J, Marinova M, Müller-Reible C, Watzka M (2008). "The vitamin K cycle". Vitam. Horm. Vitamins & Hormones. 78: 35–62. doi:10.1016/S0083-6729(07)00003-9. ISBN 9780123741134. PMID 18374189.
- Berkner KL (2005). "The vitamin K-dependent carboxylase". Annu. Rev. Nutr. 25 (1): 127–49. doi:10.1146/annurev.nutr.25.050304.092713. PMID 16011462.
- Zhang B, Ginsburg D (September 2004). "Familial multiple coagulation factor deficiencies: new biologic insight from rare genetic bleeding disorders". J. Thromb. Haemost. 2 (9): 1564–72. doi:10.1111/j.1538-7836.2004.00857.x. hdl:2027.42/74529. PMID 15333032. S2CID 7437035.
- Wallin R, Hutson SM (July 2004). "Warfarin and the vitamin K-dependent gamma-carboxylation system". Trends Mol Med. 10 (7): 299–302. doi:10.1016/j.molmed.2004.05.003. PMID 15242675.
- Berkner KL (August 2000). "The vitamin K-dependent carboxylase". J. Nutr. 130 (8): 1877–80. doi:10.1093/jn/130.8.1877. PMID 10917896.
- Presnell SR, Stafford DW (June 2002). "The vitamin K-dependent carboxylase". Thromb. Haemost. 87 (6): 937–46. doi:10.1055/s-0037-1613115. PMID 12083499. S2CID 27634025.
- Bender, David A. (2003). Nutritional biochemistry of the vitamins. Cambridge, UK: Cambridge University Press. ISBN 0-521-80388-8.
- Ball, George E. (2004). Vitamins: their role in the human body. Oxford: Blackwell Science. ISBN 0-632-06478-1.
- Combs, Gerald F. (1998). The vitamins: fundamental aspects in nutrition and health. Boston: Academic Press. ISBN 0-12-183492-1.
External links
[edit]- glutamyl+carboxylase at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
This article incorporates text from the United States National Library of Medicine, which is in the public domain.