2-Ethoxyethanol
Appearance
(Redirected from Ethoxyethanol)
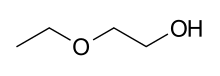 | |
| Names | |
|---|---|
| Preferred IUPAC name
2-Ethoxyethanol | |
| Other names
Cellosolve
ethylene glycol ethyl ether oxitol Ethyl Cellosolve EGEE | |
| Identifiers | |
3D model (JSmol)
|
|
| 1098271 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.003.459 |
| EC Number |
|
| 82142 | |
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1171 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H10O2 | |
| Molar mass | 90.122 g·mol−1 |
| Appearance | clear liquid |
| Odor | sweet, ether-like |
| Density | 0.930 g/cm3, liquid |
| Melting point | −70 °C (−94 °F; 203 K) |
| Boiling point | 135 °C (275 °F; 408 K) |
| miscible | |
| Vapor pressure | 4 mmHg (20°C)[1] |
| Hazards | |
| GHS labelling: | |
   
| |
| Danger | |
| H226, H302, H331, H360 | |
| P201, P202, P210, P233, P240, P241, P242, P243, P261, P264, P270, P271, P280, P281, P301+P312, P303+P361+P353, P304+P340, P308+P313, P311, P321, P330, P370+P378, P403+P233, P403+P235, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | 44 °C (111 °F; 317 K) |
| Explosive limits | 1.7%-15.6%[1] |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
2451 mg/kg (mouse, oral) 2125 mg/kg (rat, oral)[2] |
LC50 (median concentration)
|
2000 ppm (rat, 7 hr) 1820 ppm (mouse, 7 hr)[2] |
LCLo (lowest published)
|
3000 ppm (guinea pig, 24 hr)[2] |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 200 ppm (740 mg/m3) [skin][1] |
REL (Recommended)
|
TWA 0.5 ppm (1.8 mg/m3) [skin][1] |
IDLH (Immediate danger)
|
500 ppm[1] |
| Related compounds | |
Related ethers
|
2-Propoxyethanol 2-Butoxyethanol |
Related compounds
|
Ethylene glycol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
2-Ethoxyethanol, also known by the trademark Ethyl cellosolve, is a solvent used widely in commercial and industrial applications. It is a clear, colorless, nearly odorless liquid that is miscible with water, ethanol, diethyl ether, acetone, and ethyl acetate.[3]
2-Ethoxyethanol is manufactured by the reaction of ethylene oxide with ethanol.
As with other glycol ethers, 2-ethoxyethanol has the useful property of being able to dissolve chemically diverse compounds. It will dissolve oils, resins, grease, waxes, nitrocellulose, and lacquers.[3] This is an ideal property as a multi-purpose cleaner, and, therefore, 2-ethoxyethanol is used in products such as varnish removers and degreasing solutions.
References
[edit]- ^ a b c d e NIOSH Pocket Guide to Chemical Hazards. "#0258". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b c "2-Ethoxyethanol". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ a b National Research Council (U.S.). Subcommittee on Spacecraft Maximum Allowable Concentrations (1996). Spacecraft maximum allowable concentrations for selected airborne contaminants. National Academies Press. p. 189. ISBN 978-0-309-05478-2. Retrieved 19 February 2012.
External links
[edit]- "Chemical Sampling Information 2-Ethoxyethanol". www.osha.gov. Archived from the original on 8 August 2014. Retrieved 4 August 2014.
- CDC - NIOSH Pocket Guide to Chemical Hazards

