Lipoprotein lipase deficiency
| Lipoprotein lipase deficiency | |
|---|---|
| Other names | LPLD; familial chylomicronemia syndrome,[1] chylomicronemia,[2]: 533 chylomicronemia syndrome,[3] familial hyperchylomicronemia, familial hyperchylomicronemia syndrome,[4] hyperlipoproteinemia type Ia.,[5] type I hyperlipoproteinemia[6] |
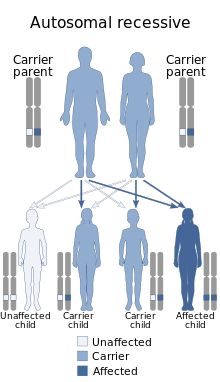 | |
| Lipoprotein lipase deficiency is inherited via autosomal recessive manner | |
| Specialty | Endocrinology |
| Causes | Genetic |
Lipoprotein lipase deficiency is a genetic disorder in which a person has a defective gene for lipoprotein lipase, which leads to very high triglycerides, which in turn causes stomach pain and deposits of fat under the skin, and which can lead to problems with the pancreas and liver, which in turn can lead to diabetes. The disorder only occurs if a child acquires the defective gene from both parents (it is autosomal recessive). It is managed by restricting fat in diet to less than 20 g/day.[7]
Signs and symptoms
[edit]The disease often presents in infancy with colicky pain, failure to thrive, and other symptoms and signs of the chylomicronemia syndrome. In women the use of estrogens or first pregnancy are also well known trigger factors for initial manifestation of LPLD. At all ages, the most common clinical manifestation is recurrent abdominal pain and acute pancreatitis. The pain may be epigastric, with radiation to the back, or it may be diffuse, with the appearance of an emergent acute abdomen. Other typical symptoms are eruptive xanthomas (in about 50% of patients), lipaemia retinalis and hepatosplenomegaly.[citation needed]
Complications
[edit]Patients with LPLD are at high risk of acute pancreatitis, which can be life-threatening, and can lead to chronic pancreatic insufficiency and diabetes.[citation needed]
Diagnosis
[edit]Lab tests show massive accumulation of chylomicrons in the plasma and corresponding severe hypertriglyceridemia. Typically, the plasma in a fasting blood sample appears creamy (plasma lactescence).[medical citation needed]
Familial LPL deficiency should be considered in anyone with severe hypertriglyceridemia and the chylomicronemia syndrome. The absence of secondary causes of severe hypertriglyceridemia (like e.g. diabetes, alcohol, estrogen-, glucocorticoid-, antidepressant- or isotretinoin-therapy, certain antihypertensive agents, and paraproteinemic disorders) increases the possibility of LPL deficiency. In this instance besides LPL also other loss-of-function mutations in genes that regulate catabolism of triglyceride-rich lipoproteins (like e.g. ApoC2, ApoA5, LMF-1, GPIHBP-1 and GPD1) should also be considered[citation needed]
The diagnosis of familial lipoprotein lipase deficiency is finally confirmed by detection of either homozygous or compound heterozygous pathogenic gene variants in LPL with either low or absent lipoprotein lipase enzyme activity.[citation needed]
Lipid measurements
· Milky, lipemic plasma revealing severe hyperchylomicronemia;[citation needed]
· Severely elevated fasting plasma triglycerides (>2000 mg/dL);[citation needed]
LPL enzyme
· Low or absent LPL activity in post-heparin plasma;[citation needed]
· LPL mass level reduced or absent in post-heparin plasma;[citation needed]
Molecular genetic testing The LPL gene is located on the short (p) arm of chromosome 8 at position 22. More than 220 mutations in the LPL gene have been found to cause familial lipoprotein lipase deficiency so far.[citation needed]
Treatment
[edit]Treatment of LPLD has two different objectives: immediate prevention of pancreatitis attacks and long-term reduction of cardiovascular disease risk.
Olezarsen (Tryngolza) was approved for medical use in the United States in December 2024.[8]
Gene therapy
[edit]In 2012, the European Commission approved alipogene tiparvovec (Glybera), a gene therapy for adults with familial LPLD (confirmed by genetic testing) and having severe or multiple pancreatitis attacks despite dietary fat restrictions. It is the first gene therapy to receive marketing authorization in the European Union; it was priced at about $1 million per treatment, and as of 2016, only one person had been treated with it commercially.[9] A total of 31 people were treated with Glybera, most for free in clinical trials before it was taken off the market.[10]
Incidence
[edit]The disorder affects about 1 out of 1,000,000 people;[11] however, epidemiological data are limited and there are regional differences due to cofounder effect (e.g. in Canada) or intermarriage.
See also
[edit]References
[edit]- ^ Santamarina-Fojo, S (1998). "Familial lipoprotein lipase deficiency". Endocrinol Metab Clin North Am. 27 (3): 551–567. doi:10.1016/S0889-8529(05)70025-6. PMID 9785052.
- ^ James, William D.; Berger, Timothy G.; et al. (2006). Andrews' Diseases of the Skin: clinical Dermatology. Saunders Elsevier. ISBN 978-0-7216-2921-6. OCLC 62736861.
- ^ Rapini, Ronald P.; Bolognia, Jean L.; Jorizzo, Joseph L. (2007). Dermatology: 2-Volume Set. St. Louis: Mosby. ISBN 978-1-4160-2999-1. OCLC 212399895.
- ^ Santamarina-Fojo, S; Brewer HB, Jr (20 February 1991). "The familial hyperchylomicronemia syndrome. New insights into underlying genetic defects". JAMA. 265 (7): 904–8. doi:10.1001/jama.1991.03460070086049. PMID 1992190.
- ^ Online Mendelian Inheritance in Man (OMIM): HYPERLIPOPROTEINEMIA, TYPE I - 238600, updated 03/18/2004. As of October 2012, mention of type Ia no longer appears in the OMIM record.
- ^ "Familial lipoprotein lipase deficiency: MedlinePlus Medical Encyclopedia". medlineplus.gov. Retrieved 17 April 2019.
- ^ Burnett, John R.; Hooper, Amanda J.; Hegele, Robert A. (June 22, 2017). "Familial Lipoprotein Lipase Deficiency". In Adam, MP; Ardinger, HH; Pagon, RA; et al. (eds.). GeneReviews. Seattle: University of Washington. PMID 20301485.
- ^ "Tryngolza (olezarsen) approved in U.S. as first-ever treatment for adults living with familial chylomicronemia syndrome as an adjunct to diet" (Press release). Ionis Pharmaceuticals. 19 December 2024. Retrieved 20 December 2024 – via PR Newswire.
- ^ Regalado, Antonio (May 4, 2016). "The World's Most Expensive Medicine Is a Bust". MIT Technology Review.
- ^ Crowe, Kelly (17 November 2018). "The million-dollar drug". CBCNews. CBC (Canadian Broadcasting Corporation). Retrieved 17 November 2018.
- ^ A.D.A.M. Editorial Board (2011-05-29). Dugdale, III, David C.; Zieve, David (eds.). Familial lipoprotein lipase deficiency. National Center for Biotechnology Information (published May 29, 2011). Retrieved October 15, 2012.
{{cite book}}:|work=ignored (help)
Further reading
[edit]- Gaudet, D; de Wal, J; Tremblay, K; Déry, S; van Deventer, S; Freidig, A; Brisson, D; Méthot, J (June 2010). "Review of the clinical development of alipogene tiparvovec gene therapy for lipoprotein lipase deficiency". Atherosclerosis. Supplements. 11 (1): 55–60. doi:10.1016/j.atherosclerosissup.2010.03.004. PMID 20427244.
- "LPL gene". NIH Genetics Home Reference. February 2015.
- Hegele, RA; et al. (August 2014). "The polygenic nature of hypertriglyceridaemia: implications for definition, diagnosis, and management". The Lancet. Diabetes & Endocrinology. 2 (8): 655–66. doi:10.1016/S2213-8587(13)70191-8. PMC 4201123. PMID 24731657.
