Fas receptor
The Fas receptor, also known as Fas, FasR, apoptosis antigen 1 (APO-1 or APT), cluster of differentiation 95 (CD95) or tumor necrosis factor receptor superfamily member 6 (TNFRSF6), is a protein that in humans is encoded by the FAS gene.[5][6] Fas was first identified using a monoclonal antibody generated by immunizing mice with the FS-7 cell line. Thus, the name Fas is derived from FS-7-associated surface antigen.[7]
The Fas receptor is a death receptor on the surface of cells that leads to programmed cell death (apoptosis) if it binds its ligand, Fas ligand (FasL). It is one of two apoptosis pathways, the other being the mitochondrial pathway.[8]
Gene
[edit]FAS receptor gene is located on the long arm of chromosome 10 (10q24.1) in humans and on chromosome 19 in mice. The gene lies on the plus (Watson strand) and is 25,255 bases in length organized into nine protein encoding exons. Similar sequences related by evolution (orthologs)[9] are found in most mammals.
Protein
[edit]Previous reports have identified as many as eight splice variants, which are translated into seven isoforms of the protein. Apoptosis-inducing Fas receptor is dubbed isoform 1 and is a type 1 transmembrane protein. Many of the other isoforms are rare haplotypes that are usually associated with a state of disease. However, two isoforms, the apoptosis-inducing membrane-bound form and the soluble form, are normal products whose production via alternative splicing is regulated by the cytotoxic RNA binding protein TIA1.[10]
The mature Fas protein has 319 amino acids, has a predicted molecular weight of 48 kilodaltons and is divided into three domains: an extracellular domain, a transmembrane domain, and a cytoplasmic domain. The extracellular domain has 157 amino acids and is rich in cysteine residues. The transmembrane and cytoplasmic domains have 17 and 145 amino acids respectively. Exons 1 through 5 encode the extracellular region. Exon 6 encodes the transmembrane region. Exons 7-9 encode the intracellular region. [citation needed]
Function
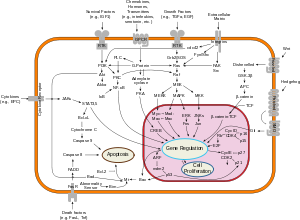
[edit]Fas forms the death-inducing signaling complex (DISC) upon ligand binding. Membrane-anchored Fas ligand trimer on the surface of an adjacent cell causes oligomerization of Fas. Recent studies which suggested the trimerization of Fas could not be validated. Other models suggested the oligomerization up to 5–7 Fas molecules in the DISC.[11] This event is also mimicked by binding of an agonistic Fas antibody, though some evidence suggests that the apoptotic signal induced by the antibody is unreliable in the study of Fas signaling. To this end, several clever ways of trimerizing the antibody for in vitro research have been employed. [citation needed]
Upon ensuing death domain (DD) aggregation, the receptor complex is internalized via the cellular endosomal machinery. This allows the adaptor molecule FADD to bind the death domain of Fas through its own death domain.[12]
FADD also contains a death effector domain (DED) near its amino terminus,[13] which facilitates binding to the DED of FADD-like interleukin-1 beta-converting enzyme (FLICE), more commonly referred to as caspase-8. FLICE can then self-activate through proteolytic cleavage into p10 and p18 subunits, two each of which form the active heterotetramer enzyme. Active caspase-8 is then released from the DISC into the cytosol, where it cleaves other effector caspases, eventually leading to DNA degradation, membrane blebbing, and other hallmarks of apoptosis. [citation needed]
Recently, Fas has also been shown to promote tumor growth, since during tumor progression, it is frequently downregulated or cells are rendered apoptosis resistant. Cancer cells in general, regardless of their Fas apoptosis sensitivity, depend on constitutive activity of Fas. This is stimulated by cancer-produced Fas ligand for optimal growth.[14]
Although Fas has been shown to promote tumor growth in the above mouse models, analysis of the human cancer genomics database revealed that FAS is not significantly focally amplified across a dataset of 3131 tumors (FAS is not an oncogene), but is significantly focally deleted across the entire dataset of these 3131 tumors,[15] suggesting that FAS functions as a tumor suppressor in humans.
In cultured cells, FasL induces various types of cancer cell apoptosis through the Fas receptor. In AOM-DSS-induced colon carcinoma and MCA-induced sarcoma mouse models, it has been shown that Fas acts as a tumor suppressor.[16] Furthermore, the Fas receptor also mediates tumor-specific cytotoxic T lymphocyte (CTL) anti-tumor cytotoxicity.[17] In addition to the well-described on-target CTL anti-tumor cytotoxicity, Fas has been ascribed with a distinct function – the induction of bystander tumor cell death even amongst cognate antigen non-expressing (bystander) cells. CTL-mediated bystander killing was described by the Fleischer Lab in 1986[18] and later attributed to fas-mediated lysis in vitro by the Austin Research Institute, Cellular Cytotoxicity Laboratory.[19] More recently, fas-mediated bystander tumor cell killing was demonstrated in vivo by the Lymphoma Immunotherapy Program at Mount Sinai School of Medicine using T cells and CAR-T cells,[20] similar to additional in vitro work using bispecific antibodies performed at Amgen.[21]
Role in apoptosis
[edit]Some reports have suggested that the extrinsic Fas pathway is sufficient to induce complete apoptosis in certain cell types through DISC assembly and subsequent caspase-8 activation. These cells are dubbed Type 1 cells and are characterized by the inability of anti-apoptotic members of the Bcl-2 family (namely Bcl-2 and Bcl-xL) to protect from Fas-mediated apoptosis. Characterized Type 1 cells include H9, CH1, SKW6.4 and SW480, all of which are lymphocyte lineages except the latter, which is a colon adenocarcinoma lineage. However, evidence for crosstalk between the extrinsic and intrinsic pathways exists in the Fas signal cascade. [citation needed]
In most cell types, caspase-8 catalyzes the cleavage of the pro-apoptotic BH3-only protein Bid into its truncated form, tBid. BH-3 only members of the Bcl-2 family exclusively engage anti-apoptotic members of the family (Bcl-2, Bcl-xL), allowing Bak and Bax to translocate to the outer mitochondrial membrane, thus permeabilizing it and facilitating release of pro-apoptotic proteins such as cytochrome c and Smac/DIABLO, an antagonist of inhibitors of apoptosis proteins (IAPs). [citation needed]
Interactions
[edit]Fas receptor has been shown to interact with:
- Caspase 8,[22][23][24]
- Caspase 10,[25]
- CFLAR,[23][24]
- FADD,[22][23][26][27][28][29]
- Fas ligand,[22][30][31][32]
- PDCD6,[33] and
- Small ubiquitin-related modifier 1.[34][35]
References
[edit]- ^ a b c GRCh38: Ensembl release 89: ENSG00000026103 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000024778 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Lichter P, Walczak H, Weitz S, Behrmann I, Krammer PH (September 1992). "The human APO-1 (APT) antigen maps to 10q23, a region that is syntenic with mouse chromosome 19". Genomics. 14 (1): 179–80. doi:10.1016/S0888-7543(05)80302-7. PMID 1385299.
- ^ Inazawa J, Itoh N, Abe T, Nagata S (November 1992). "Assignment of the human Fas antigen gene (Fas) to 10q24.1". Genomics. 14 (3): 821–2. doi:10.1016/S0888-7543(05)80200-9. PMID 1385309.
- ^ Nagata S (July 2004). "Early work on the function of CD95, an interview with Shige Nagata". Cell Death and Differentiation. 11 (Suppl 1): S23-7. doi:10.1038/sj.cdd.4401453. PMID 15143352.
- ^ Wajant H (May 2002). "The Fas signaling pathway: more than a paradigm". Science. 296 (5573): 1635–6. Bibcode:2002Sci...296.1635W. doi:10.1126/science.1071553. PMID 12040174. S2CID 29449108.
- ^ "OrthoMaM phylogenetic marker: FAS coding sequence". Archived from the original on 2016-03-03. Retrieved 2009-12-02.
- ^ Izquierdo JM, Majós N, Bonnal S, Martínez C, Castelo R, Guigó R, et al. (August 2005). "Regulation of Fas alternative splicing by antagonistic effects of TIA-1 and PTB on exon definition". Molecular Cell. 19 (4): 475–84. doi:10.1016/j.molcel.2005.06.015. PMID 16109372.
- ^ Wang L, Yang JK, Kabaleeswaran V, Rice AJ, Cruz AC, Park AY, et al. (November 2010). "The Fas-FADD death domain complex structure reveals the basis of DISC assembly and disease mutations". Nature Structural & Molecular Biology. 17 (11): 1324–9. doi:10.1038/nsmb.1920. PMC 2988912. PMID 20935634.
- ^ Huang B, Eberstadt M, Olejniczak ET, Meadows RP, Fesik SW (1996). "NMR structure and mutagenesis of the Fas (APO-1/CD95) death domain". Nature. 384 (6610): 638–41. Bibcode:1996Natur.384..638H. doi:10.1038/384638a0. PMID 8967952. S2CID 2492303.
- ^ Eberstadt M, Huang B, Chen Z, Meadows RP, Ng SC, Zheng L, et al. (April 1998). "NMR structure and mutagenesis of the FADD (Mort1) death-effector domain". Nature. 392 (6679): 941–5. Bibcode:1998Natur.392..941E. doi:10.1038/31972. PMID 9582077. S2CID 4370202.
- ^ Chen L, Park SM, Tumanov AV, Hau A, Sawada K, Feig C, et al. (May 2010). "CD95 promotes tumour growth". Nature. 465 (7297): 492–6. Bibcode:2010Natur.465..492C. doi:10.1038/nature09075. PMC 2879093. PMID 20505730.
- ^ "Tumorscape". The Broad Institute. Archived from the original on 2012-04-14. Retrieved 2012-07-05.
- ^ Liu F, Bardhan K, Yang D, Thangaraju M, Ganapathy V, Waller JL, et al. (July 2012). "NF-κB directly regulates Fas transcription to modulate Fas-mediated apoptosis and tumor suppression". The Journal of Biological Chemistry. 287 (30): 25530–40. doi:10.1074/jbc.M112.356279. PMC 3408167. PMID 22669972.
- ^ Yang D, Torres CM, Bardhan K, Zimmerman M, McGaha TL, Liu K (May 2012). "Decitabine and vorinostat cooperate to sensitize colon carcinoma cells to Fas ligand-induced apoptosis in vitro and tumor suppression in vivo". Journal of Immunology. 188 (9): 4441–9. doi:10.4049/jimmunol.1103035. PMC 3398838. PMID 22461695.
- ^ Fleischer B (August 1986). "Lysis of bystander target cells after triggering of human cytotoxic T lymphocytes". European Journal of Immunology. 16 (8): 1021–4. doi:10.1002/eji.1830160826. PMID 3488908. S2CID 27562316.
- ^ Smyth MJ, Krasovskis E, Johnstone RW (July 1998). "Fas ligand-mediated lysis of self bystander targets by human papillomavirus-specific CD8+ cytotoxic T lymphocytes". Journal of Virology. 72 (7): 5948–54. doi:10.1128/JVI.72.7.5948-5954.1998. PMC 110399. PMID 9621057.
- ^ Upadhyay R, Boiarsky JA, Pantsulaia G, Svensson-Arvelund J, Lin MJ, Wroblewska A, et al. (December 2020). "A critical role for fas-mediated off-target tumor killing in T cell immunotherapy". Cancer Discovery. 11 (3): 599–613. doi:10.1158/2159-8290.CD-20-0756. ISSN 2159-8274. PMC 7933082. PMID 33334730.
- ^ Ross SL, Sherman M, McElroy PL, Lofgren JA, Moody G, Baeuerle PA, et al. (2017-08-24). "Bispecific T cell engager (BiTE®) antibody constructs can mediate bystander tumor cell killing". PLOS ONE. 12 (8): e0183390. Bibcode:2017PLoSO..1283390R. doi:10.1371/journal.pone.0183390. PMC 5570333. PMID 28837681.
- ^ a b c Gajate C, Mollinedo F (March 2005). "Cytoskeleton-mediated death receptor and ligand concentration in lipid rafts forms apoptosis-promoting clusters in cancer chemotherapy". The Journal of Biological Chemistry. 280 (12): 11641–7. doi:10.1074/jbc.M411781200. PMID 15659383.
- ^ a b c MacFarlane M, Ahmad M, Srinivasula SM, Fernandes-Alnemri T, Cohen GM, Alnemri ES (October 1997). "Identification and molecular cloning of two novel receptors for the cytotoxic ligand TRAIL". The Journal of Biological Chemistry. 272 (41): 25417–20. doi:10.1074/jbc.272.41.25417. PMID 9325248.
- ^ a b Shu HB, Halpin DR, Goeddel DV (June 1997). "Casper is a FADD- and caspase-related inducer of apoptosis". Immunity. 6 (6): 751–63. doi:10.1016/S1074-7613(00)80450-1. PMID 9208847.
- ^ Vincenz C, Dixit VM (March 1997). "Fas-associated death domain protein interleukin-1beta-converting enzyme 2 (FLICE2), an ICE/Ced-3 homologue, is proximally involved in CD95- and p55-mediated death signaling". The Journal of Biological Chemistry. 272 (10): 6578–83. doi:10.1074/jbc.272.10.6578. PMID 9045686.
- ^ Pan G, O'Rourke K, Chinnaiyan AM, Gentz R, Ebner R, Ni J, Dixit VM (April 1997). "The receptor for the cytotoxic ligand TRAIL". Science. 276 (5309): 111–3. doi:10.1126/science.276.5309.111. PMID 9082980. S2CID 19984057.
- ^ Huang B, Eberstadt M, Olejniczak ET, Meadows RP, Fesik SW (1996). "NMR structure and mutagenesis of the Fas (APO-1/CD95) death domain". Nature. 384 (6610): 638–41. Bibcode:1996Natur.384..638H. doi:10.1038/384638a0. PMID 8967952. S2CID 2492303.
- ^ Chinnaiyan AM, O'Rourke K, Tewari M, Dixit VM (May 1995). "FADD, a novel death domain-containing protein, interacts with the death domain of Fas and initiates apoptosis". Cell. 81 (4): 505–12. doi:10.1016/0092-8674(95)90071-3. PMID 7538907. S2CID 16906755.
- ^ Thomas LR, Stillman DJ, Thorburn A (September 2002). "Regulation of Fas-associated death domain interactions by the death effector domain identified by a modified reverse two-hybrid screen". The Journal of Biological Chemistry. 277 (37): 34343–8. doi:10.1074/jbc.M204169200. PMID 12107169.
- ^ Micheau O, Tschopp J (July 2003). "Induction of TNF receptor I-mediated apoptosis via two sequential signaling complexes" (PDF). Cell. 114 (2): 181–90. doi:10.1016/S0092-8674(03)00521-X. PMID 12887920. S2CID 17145731.
- ^ Starling GC, Bajorath J, Emswiler J, Ledbetter JA, Aruffo A, Kiener PA (April 1997). "Identification of amino acid residues important for ligand binding to Fas". The Journal of Experimental Medicine. 185 (8): 1487–92. doi:10.1084/jem.185.8.1487. PMC 2196280. PMID 9126929.
- ^ Schneider P, Bodmer JL, Holler N, Mattmann C, Scuderi P, Terskikh A, et al. (July 1997). "Characterization of Fas (Apo-1, CD95)-Fas ligand interaction". The Journal of Biological Chemistry. 272 (30): 18827–33. doi:10.1074/jbc.272.30.18827. PMID 9228058.
- ^ Jung YS, Kim KS, Kim KD, Lim JS, Kim JW, Kim E (October 2001). "Apoptosis-linked gene 2 binds to the death domain of Fas and dissociates from Fas during Fas-mediated apoptosis in Jurkat cells". Biochemical and Biophysical Research Communications. 288 (2): 420–6. doi:10.1006/bbrc.2001.5769. PMID 11606059.
- ^ Okura T, Gong L, Kamitani T, Wada T, Okura I, Wei CF, et al. (November 1996). "Protection against Fas/APO-1- and tumor necrosis factor-mediated cell death by a novel protein, sentrin". Journal of Immunology. 157 (10): 4277–81. doi:10.4049/jimmunol.157.10.4277. PMID 8906799. S2CID 38606511.
- ^ Ryu SW, Chae SK, Kim E (December 2000). "Interaction of Daxx, a Fas binding protein, with sentrin and Ubc9". Biochemical and Biophysical Research Communications. 279 (1): 6–10. doi:10.1006/bbrc.2000.3882. PMID 11112409.
Further reading
[edit]- Nagata S (February 1997). "Apoptosis by death factor". Cell. 88 (3): 355–65. doi:10.1016/S0092-8674(00)81874-7. PMID 9039262. S2CID 494841.
- Cascino I, Papoff G, Eramo A, Ruberti G (January 1996). "Soluble Fas/Apo-1 splicing variants and apoptosis". Frontiers in Bioscience. 1 (4): d12-8. doi:10.2741/A112. PMID 9159204.
- Uckun FM (September 1998). "Bruton's tyrosine kinase (BTK) as a dual-function regulator of apoptosis". Biochemical Pharmacology. 56 (6): 683–91. doi:10.1016/S0006-2952(98)00122-1. PMID 9751072.
- Krammer PH (October 2000). "CD95's deadly mission in the immune system". Nature. 407 (6805): 789–95. Bibcode:2000Natur.407..789K. doi:10.1038/35037728. PMID 11048730. S2CID 4328897.
- Siegel RM, Chan FK, Chun HJ, Lenardo MJ (December 2000). "The multifaceted role of Fas signaling in immune cell homeostasis and autoimmunity". Nature Immunology. 1 (6): 469–74. doi:10.1038/82712. PMID 11101867. S2CID 345769.
- Yonehara S (2003). "Death receptor Fas and autoimmune disease: from the original generation to therapeutic application of agonistic anti-Fas monoclonal antibody". Cytokine & Growth Factor Reviews. 13 (4–5): 393–402. doi:10.1016/S1359-6101(02)00024-2. PMID 12220552.
- Choi C, Benveniste EN (January 2004). "Fas ligand/Fas system in the brain: regulator of immune and apoptotic responses". Brain Research. Brain Research Reviews. 44 (1): 65–81. doi:10.1016/j.brainresrev.2003.08.007. PMID 14739003. S2CID 46587211.
- Poppema S, Maggio E, van den Berg A (March 2004). "Development of lymphoma in Autoimmune Lymphoproliferative Syndrome (ALPS) and its relationship to Fas gene mutations". Leukemia & Lymphoma. 45 (3): 423–31. doi:10.1080/10428190310001593166. PMID 15160902. S2CID 35128360.
External links
[edit]- FAS+Receptor at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Overview of all the structural information available in the PDB for UniProt: P25445 (Human Tumor necrosis factor receptor superfamily member 6) at the PDBe-KB.
- Overview of all the structural information available in the PDB for UniProt: P25446 (Mouse Tumor necrosis factor receptor superfamily member 6) at the PDBe-KB.